 V10 Battery Replacement 25.2V 6000mAh for Dyson V10 Series
V10 Battery Replacement 25.2V 6000mAh for Dyson V10 Series
 Zengyu New Battery Replace Bdc72 for Total Station OS / Es
Zengyu New Battery Replace Bdc72 for Total Station OS / Es
 Best Performance Surgical Power Drill Total Hip Replacement
Best Performance Surgical Power Drill Total Hip Replacement
 Medical Drill Tools Equipment Orthopedic Low-Speed Endoprosthesis
Medical Drill Tools Equipment Orthopedic Low-Speed Endoprosthesis
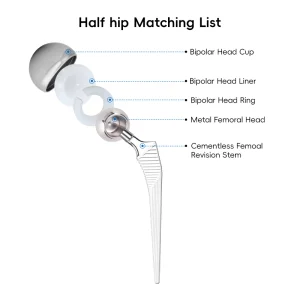
 Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
 Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Replacement
Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Replacement
 Total Hip Replacement for Osteoarthritis Secondary to Hip Dysplasia
Total Hip Replacement for Osteoarthritis Secondary to Hip Dysplasia
 Zengyu New Type 109000 Charger to Replace for Trimble Two 2 Slots
Zengyu New Type 109000 Charger to Replace for Trimble Two 2 Slots
Total Hip Arthroplasty (THA), commonly known as Total Hip Replacement (THR), has emerged as one of the most successful and cost-effective surgical procedures in modern medicine. As a critical sector for Total Hip Replacement Manufacturers, the global market is witnessing an unprecedented surge. The primary drivers include a rapidly aging global population, the rising prevalence of arthritis, and a significant increase in sports-related injuries among younger demographics.
Industrial data indicates that the orthopedic implants market is expanding at a CAGR of over 5.5%. For an Exporter of THR systems, this represents a vast opportunity. Hospitals in the USA, Europe, and emerging economies in Asia are shifting towards minimally invasive surgeries that require high-precision implants—products that ensure faster recovery and longer prosthetic lifespans.
The transition from traditional titanium alloys to advanced biocompatible ceramics and 3D-printed porous surfaces is redefining the industry. As a leading manufacturer, we are integrating AI-assisted design and robotic manufacturing to produce implants that mimic the natural human anatomy with micron-level accuracy.
Enhancing osseointegration by creating structures that mimic human trabecular bone, allowing for faster biological fixation.
Designing instrument sets compatible with robotic surgical systems to ensure perfect cup positioning and leg length restoration.
Utilizing highly cross-linked polyethylene to drastically reduce wear rates and extend the life of the implant for younger patients.
Procurement teams at major healthcare groups and national health ministries look for more than just a product; they seek a reliable supply chain partner. Our role as an exporter involves navigating complex regulatory environments, ensuring that every shipment meets the localized medical standards of the destination country, whether it's the CE marking for Europe or specific FDA requirements for the Americas.
In localized scenarios, such as specialized orthopedic clinics in Southeast Asia or large-scale public hospitals in Latin America, the demand ranges from standard cementless systems to complex revision sets. We provide the full spectrum of necessary tools, from surgical drills to femoral heads, ensuring a turnkey solution for surgeons.

We are a GMP and ISO certified global orthopedic implants manufacturing company, committed to designing and manufacturing products that are safe, effective, and of high quality. Our products meet both National and International Standards, ensuring patients receive the best care possible.
With over 18+ years of experience as a leading orthopedic implants manufacturer and exporter, we have successfully expanded our footprint to 75+ countries. Our Shanghai-based operations, Shanghai XOVEC Medical Instrument Co., Ltd., ensure that every innovation undergoes complete monitoring and seamless documentation from procurement to supply.
Our mission is simple: to provide reliable, safe, and affordable Orthopedic implants of high geometric accuracy and surface quality. We prioritize long service life and biocompatibility, aiming for fast healing success for patients worldwide.

A trusted entity with official recognition, ensuring adherence to strict legal and medical protocols.

Delivering high-quality products that balance superior performance with global market affordability.

A proven track record of international logistics and regulatory compliance across diverse markets.

Flexible manufacturing capabilities to meet specific regional requirements and surgical preferences.
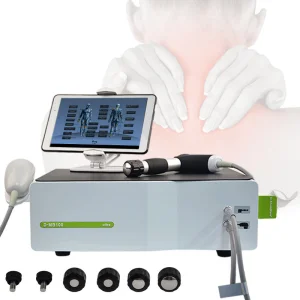 2 in 1 Shockwave Vibration Sw13 Replacement Therapy
2 in 1 Shockwave Vibration Sw13 Replacement Therapy
 Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Instrument Set
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Instrument Set
 China Factory OEM Support Thr/Tkr Total Joint Replacement Instrument
China Factory OEM Support Thr/Tkr Total Joint Replacement Instrument
 Medical Instrument Orthopedic Electric Bone Saw for Thr
Medical Instrument Orthopedic Electric Bone Saw for Thr
 Ss-1011 Orthopedic Thr Total Hip Replacement Oscillating Surgical Saw
Ss-1011 Orthopedic Thr Total Hip Replacement Oscillating Surgical Saw
 Surgical Veterinary Thr Dog Total Hip Replacement Joint
Surgical Veterinary Thr Dog Total Hip Replacement Joint
 Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
 Hot Sale Replacement 7.4V 2600mAh Geb212 Li Ion Battery
Hot Sale Replacement 7.4V 2600mAh Geb212 Li Ion Battery
Modern Total Hip Replacement (THR) is a marvel of bio-engineering. As leading manufacturers, we focus on four primary components: the acetabular cup, the liner (often made of cross-linked polyethylene or ceramic), the femoral head (cobalt-chrome or ceramic), and the femoral stem. Each of these requires a different manufacturing process. The stems are often forged or CNC-milled from Ti-6Al-4V alloy, known for its incredible strength-to-weight ratio and biocompatibility.
A significant portion of our global exports targets the treatment of Osteoarthritis secondary to hip dysplasia. These complex cases require specialized femoral head systems and revision instruments. By providing a wide range of sizes and modular components, we allow surgeons to customize the implant to the patient's unique bone morphology, drastically reducing the risk of post-operative luxation.
The trend toward sustainability is even reaching the medical sector. As a responsible China-based factory, we are optimizing our production lines to reduce waste in titanium milling. Our sterilized packaging is also evolving to meet "green" healthcare initiatives without sacrificing the essential barrier properties required for a 5-year shelf life.
One of the critical factors for THR longevity is "Primary Fixation." Our cementless implants feature plasma-sprayed hydroxyapatite (HA) coatings. This bio-active material encourages the patient's own bone to grow directly onto the implant, creating a permanent bond that eliminates the need for bone cement and minimizes the risk of aseptic loosening, which is the leading cause of late-stage revision surgery.