













| Still deciding? Get samples of $ ! US$ 413/Piece Request Sample |
| Model NO. | 2303.2410201 | Material | Titanium |
| Feature | Disposable | Certification | CE, FDA, ISO13485 |
| Group | Adult & Children | Product Name | Non-Absorbable Suture Anchor |
| MOQ | 1 Piece | OEM | Available |
| Origin | China | HS Code | 9021100090 |


Modern implants utilize advanced bearing surfaces like ceramic-on-ceramic or ceramic-on-polyethylene, which are highly wear-resistant. This greatly extends the implant's lifespan and reduces the long-term risk of revision surgery.
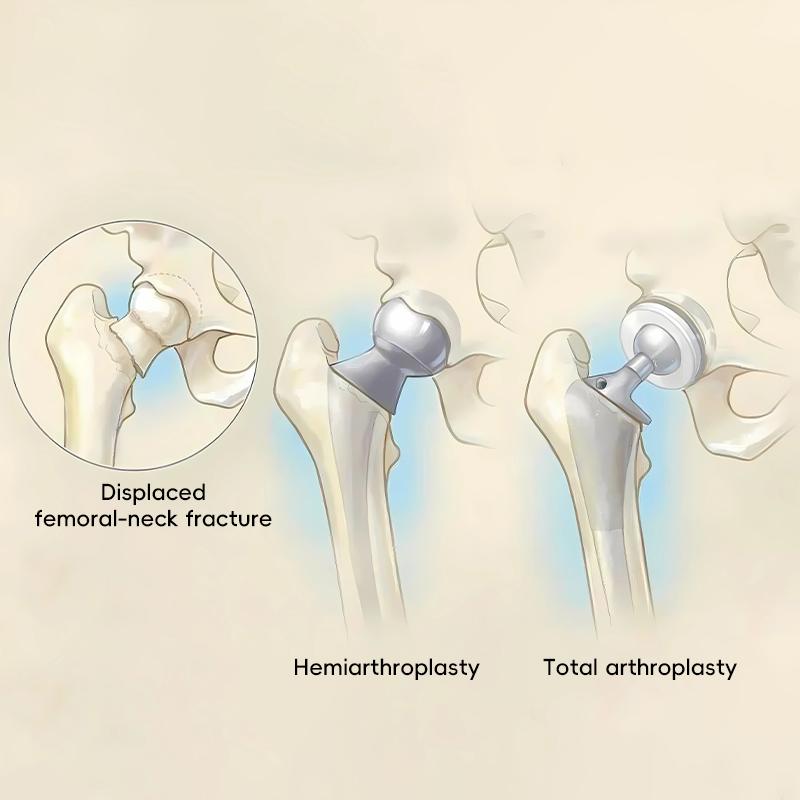
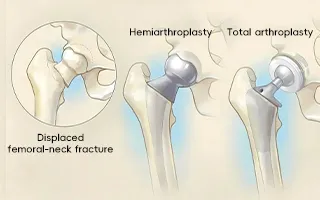
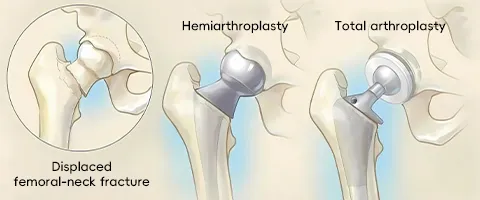
The product portfolio covers both hemiarthroplasty (replacing only the femoral head) and total hip replacement (replacing both the femoral head and acetabulum), providing surgeons with maximum choice.

Through a modular model design system, surgeons can tailor the procedure for each patient like assembling parts for a custom fit.
Each product has a clear matching list that strictly defines which models of femoral heads, stems, acetabular cups, and liners can be safely combined.


| Component | Technical Introduction |
|---|---|
| Acetabular Cup | Implanted into the pelvis to replace damaged cartilage. Typically porous to encourage bone growth (osseointegration). Made of titanium or cobalt-chromium alloy. |
| Acetabular Liner | A wear-resistant insert fitted inside the metal cup. Serves as the actual bearing surface. Made from highly cross-linked polyethylene, ceramic, or metal. |
| Femoral Head | Spherical ball component that rotates within the liner. Enables range of motion (flexion, rotation). Made of ceramic or polished metal. |
| Femoral Stem | Inserted into the thigh bone for stability. Features a tapered connection for the head. May have coatings for bone ingrowth. |
As a leader in orthopedic implants and instruments manufacturing, we have been successfully supplying to 2,500+ clients in 70+ countries for over 15 years.

Comprehensive product portfolio spanning orthopedics, trauma care, and veterinary medicine.
1-year warranty provided. Technical responses and complaints are addressed within 2 days.
We are CE certified and ISO 13485 quality management system certified.



 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants