

1 / 5










| Still deciding? Get samples of $ ! US$ 119/Piece Request Sample |






| REF | Description |
|---|---|
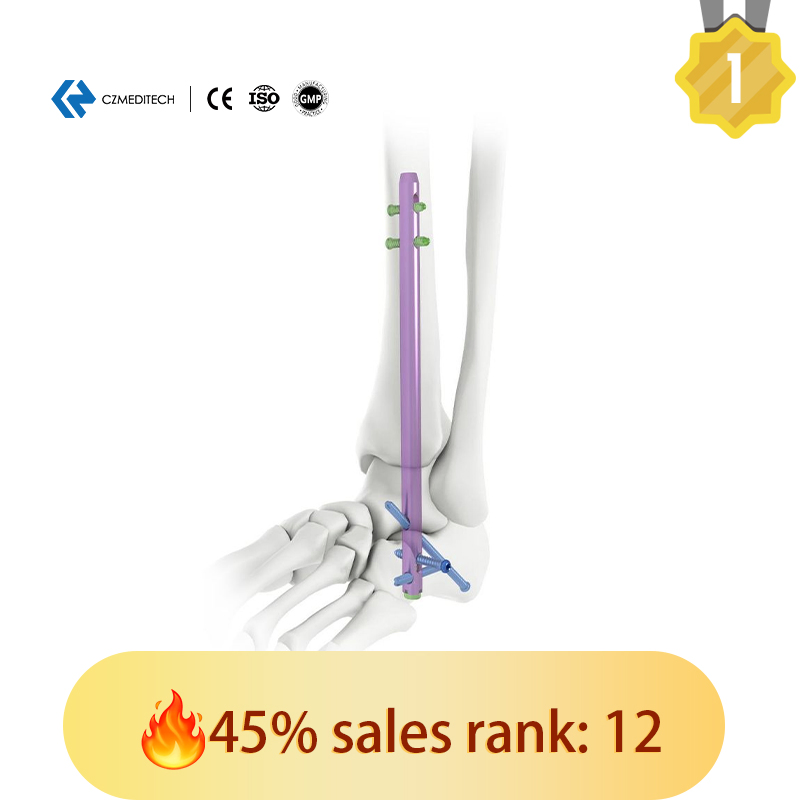
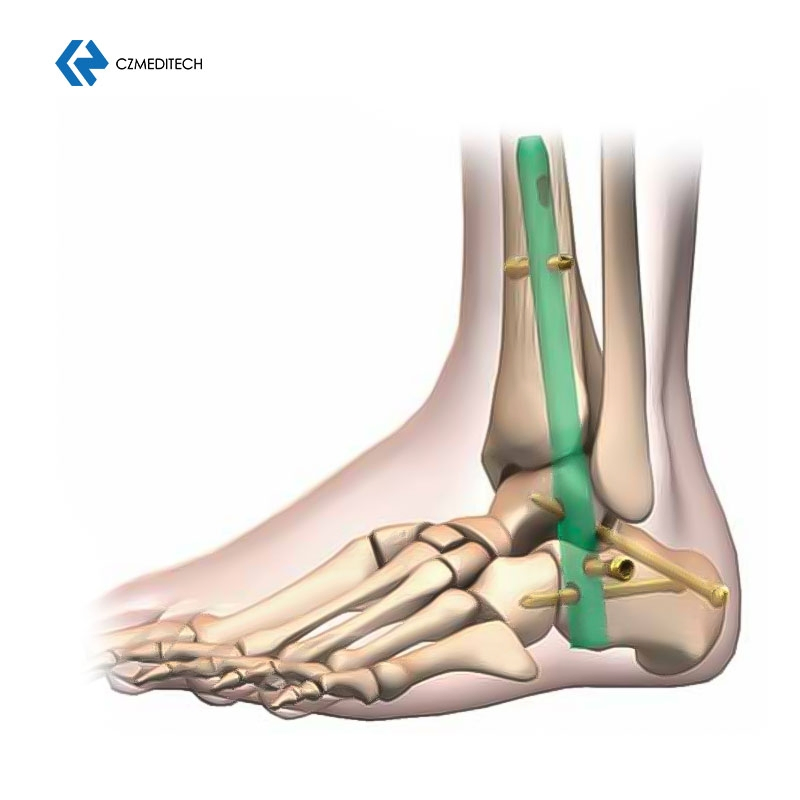
| Hindfoot Fusion Ankle Nail | 10*150/200/225/250 mm (L) |
| 10*150/200 mm (R) | |
| 11*150/200/250 mm (L) | |
| 11*150/200/225/250 mm (R) | |
| 12*150/200/250 mm (L) | |
| 12*150/200/250 mm (R) | |
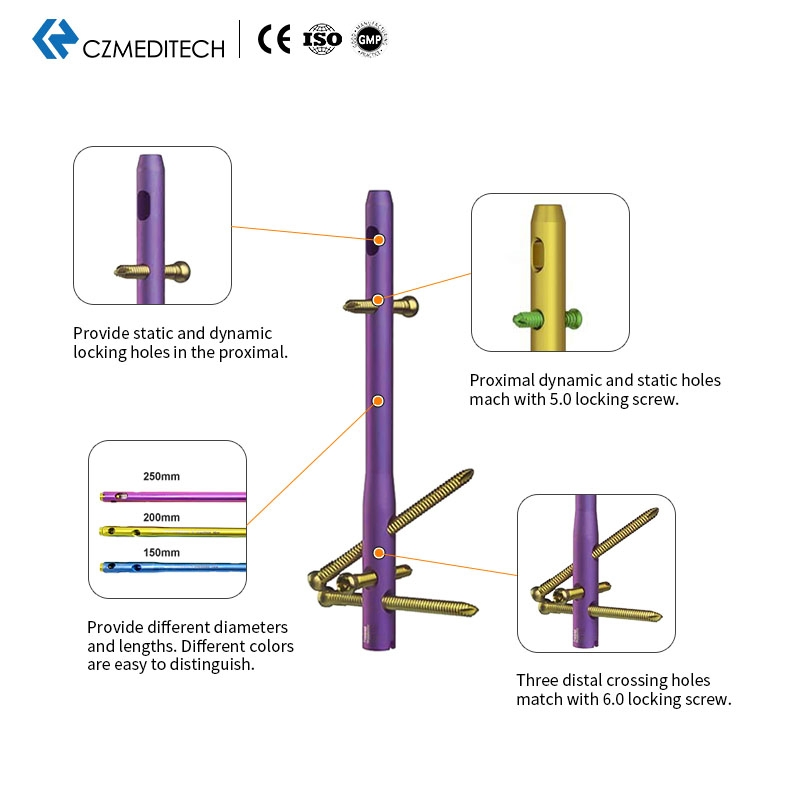
| 5.0mm Locking Screws | 5.0*28/30/32.../60mm |
| 6.0mm Locking Screws | 6.0*26/28/30.../100mm |
As a leader in orthopedic implants and instruments manufacturing, we have successfully supplied to 2,500+ clients in 70+ countries for over 15 years.

A comprehensive product portfolio spanning orthopedics, trauma care, veterinary medicine, and sterilization.

We provide a 1-year warranty. All complaints receive responses within 2 days.
We are CE certified and ISO 13485 quality management system certified.



 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants