


1 / 5










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |

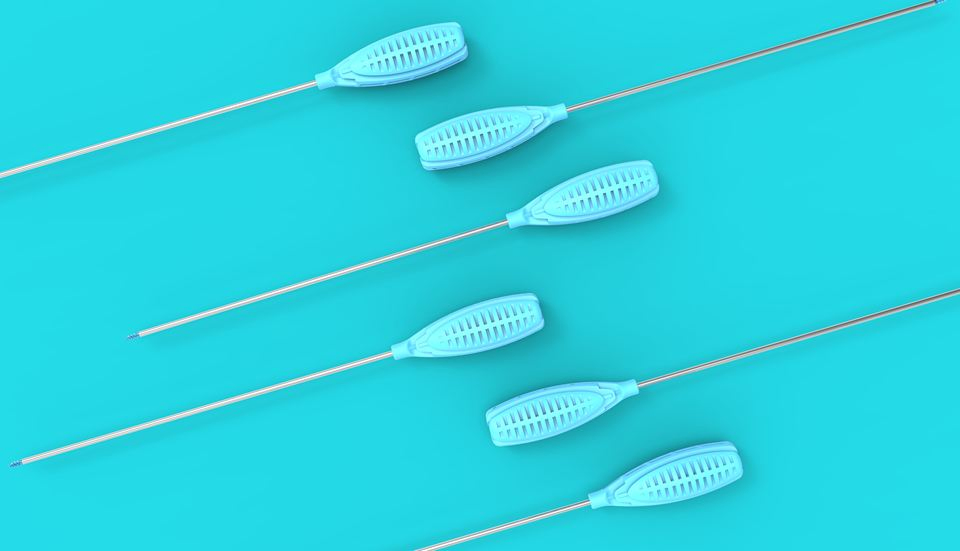
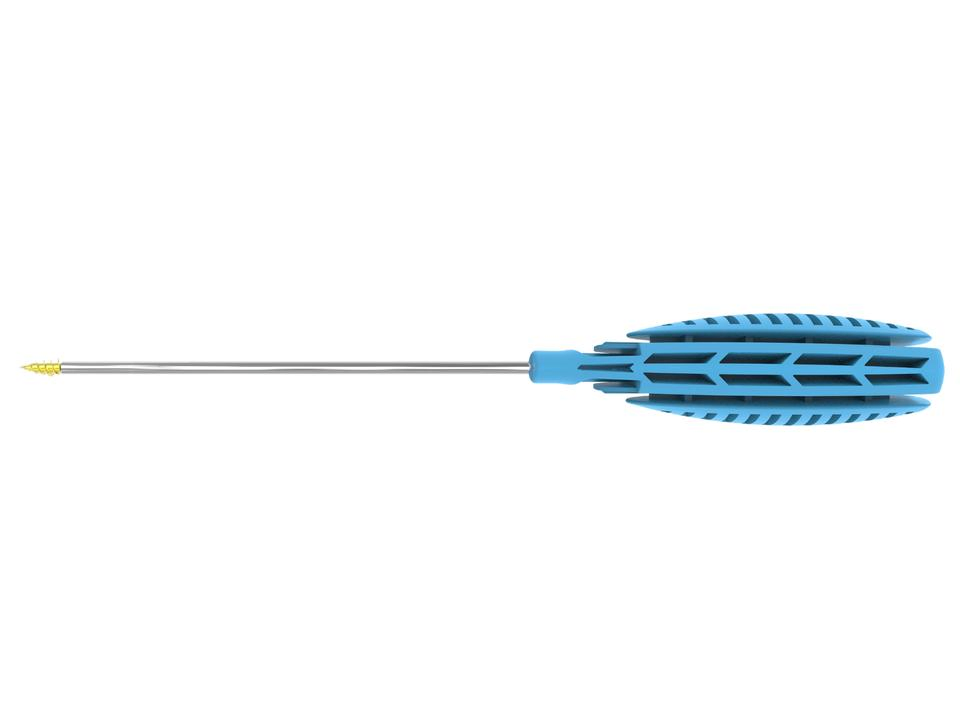
| Product Name | Specification | Anchor Outer Diameter (mm) | Anchor Length (mm) | Total Length (mm) | Suture Spec |
|---|---|---|---|---|---|
| Suture Anchor For Joint Tendon Repair | T12-φ2.0×7.5 | φ2.0 | 7.5 | 184.3 | 2 #0 |
| T12-φ2.8×11.5 | φ2.8 | 11.5 | 251.4 | 1 #2 | |
| T12-φ3.5×12 | φ3.5 | 12 | 255.2 | 2 #2 | |
| T12-φ5.0×15.5 | φ5.0 | 15.5 | 257.5 | 2 #2 | |
| T12-φ6.5×16.3 | φ6.5 | 16.3 | 260.7 | 2 #2 |
*All prices are for reference only. Exact price based on latest quotation.




 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants