












| Still deciding? Get samples of $ ! US$ 600/Piece Request Sample |
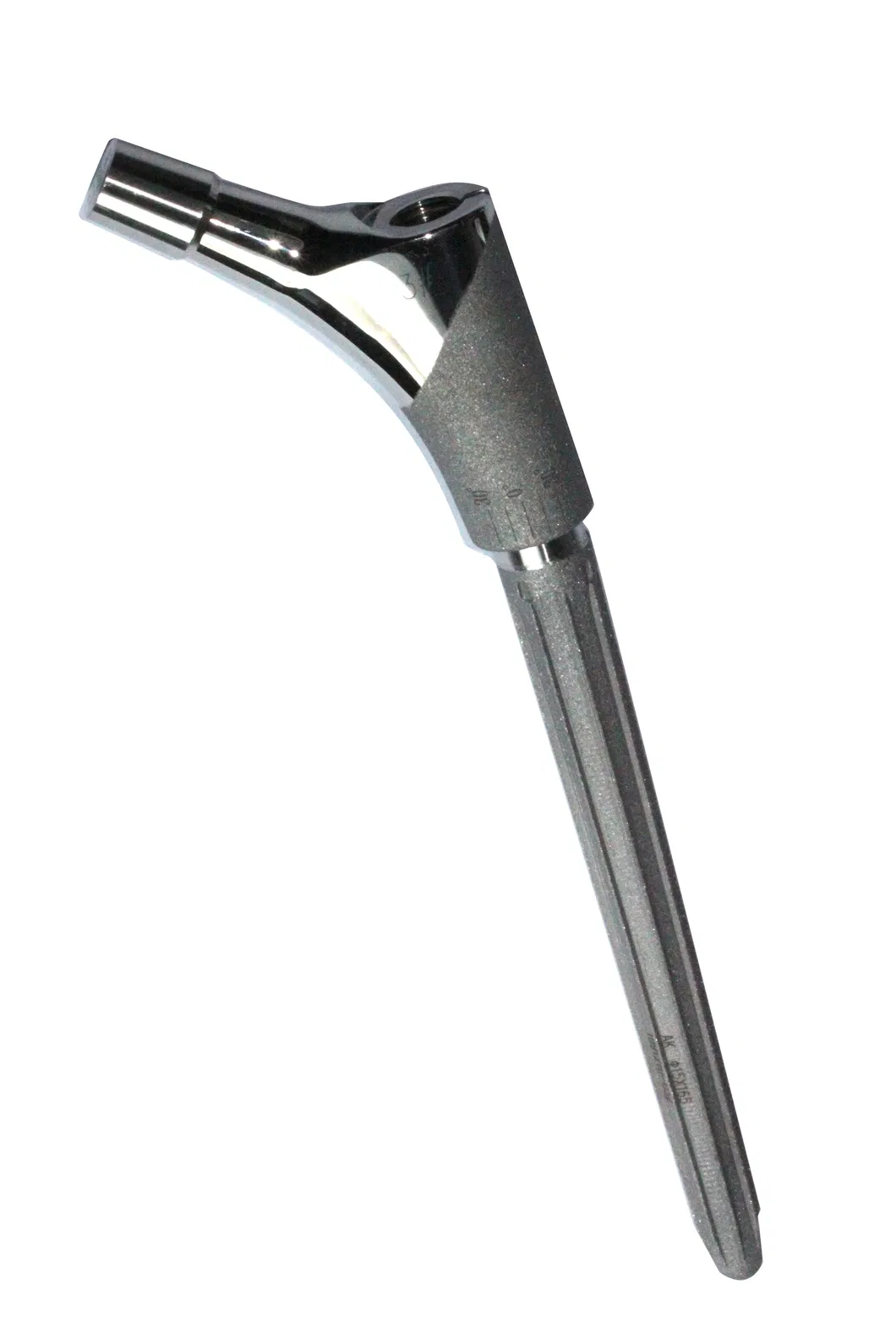
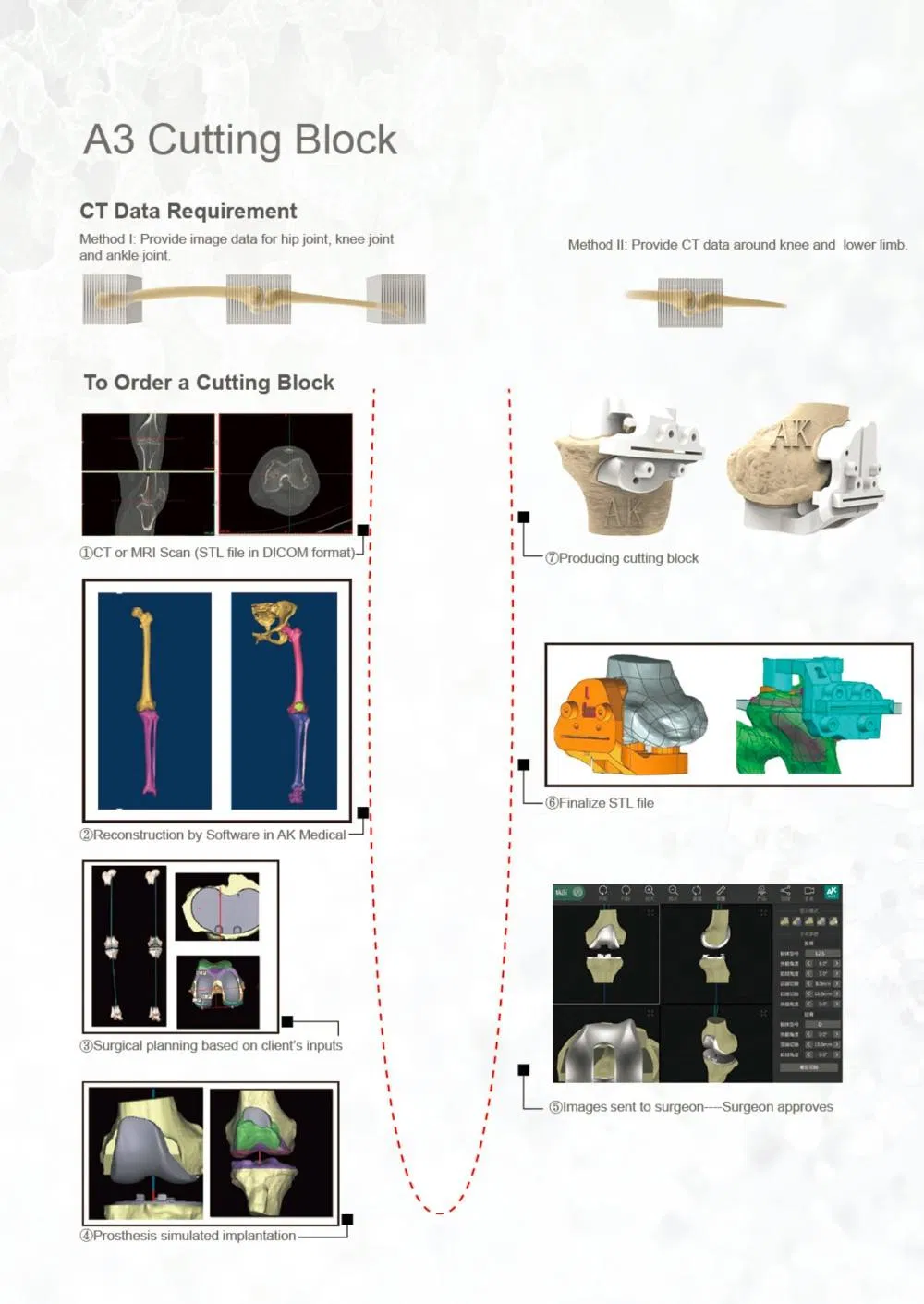
3D ACT series products achieve the innovative breakthrough in personalized medical solutions, the world's first series products that get the clinical approval to sell in market.
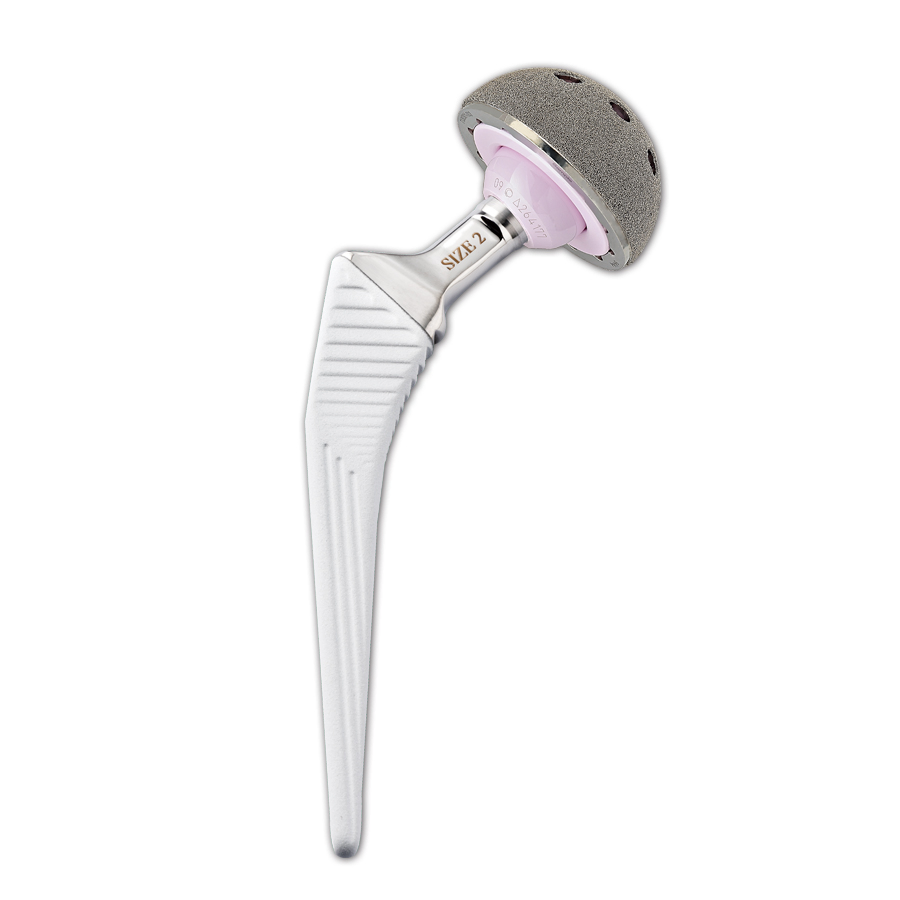
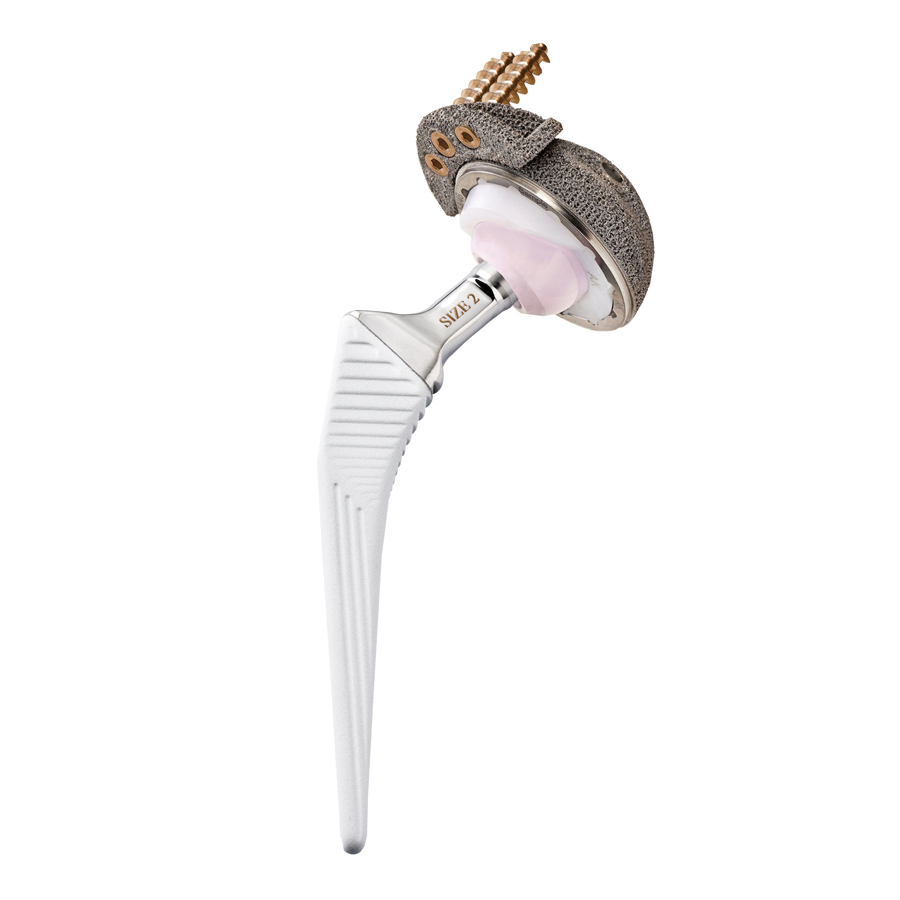
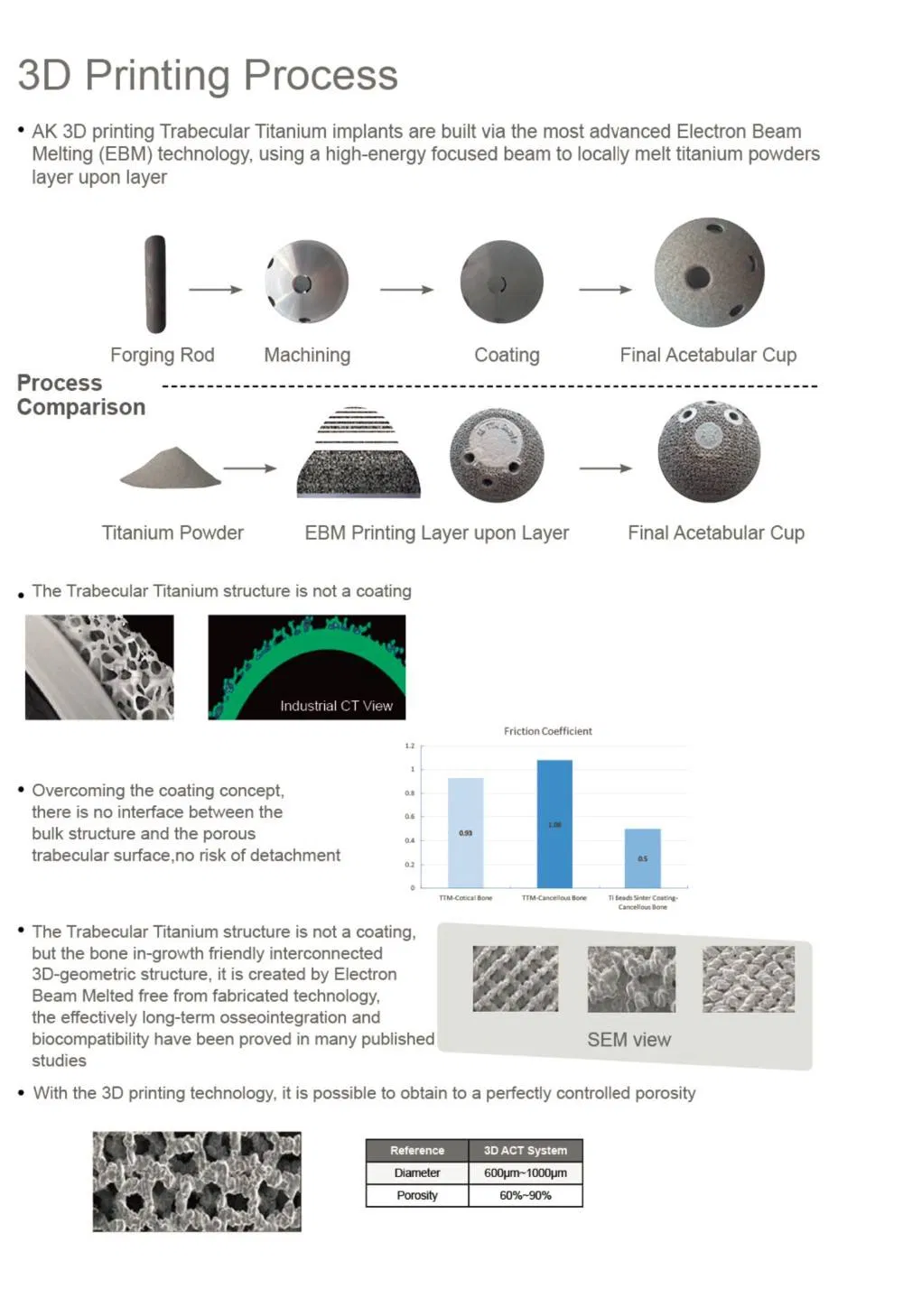
The prosthetic components in Trabecular Titanium are built via the most advanced Electron Beam Melting (EBM) technology, using a high-energy focused beam to locally melt metallic powders layer upon layer. Overcoming the coating concept, there is no interface between the bulk structure and the porous trabecular surface.
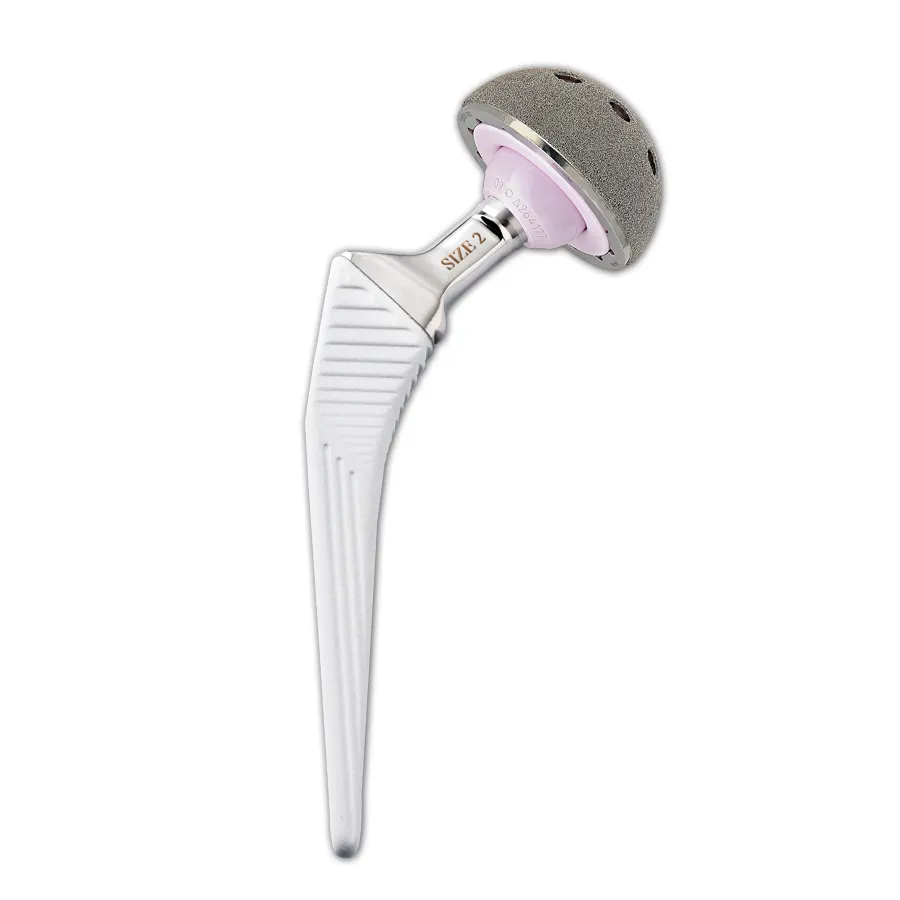


The regular, three-dimensional, hexagonal cell structure imitates trabecular bone morphology; it has a high open porosity (80%) and a mean pore diameter of 800 μm. High open porosity and adequate pore size are expected to enhance cell migration and vascularization, favoring new bone formation.


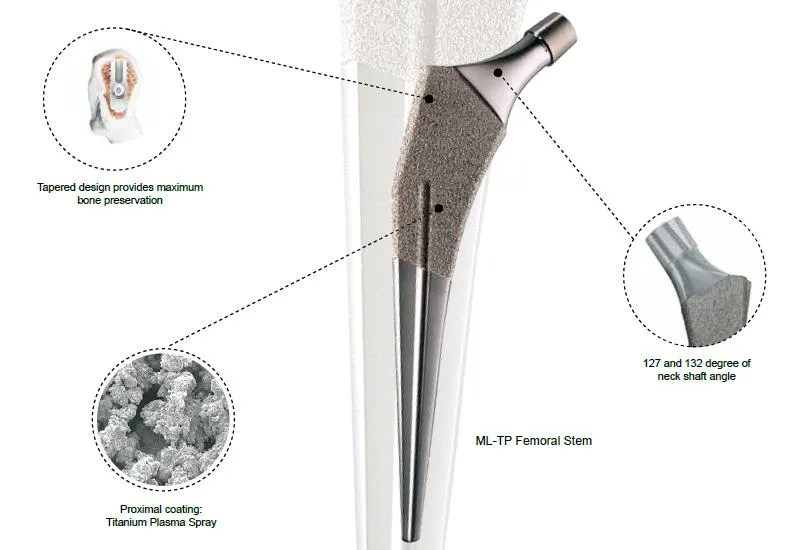

 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants