













| Still deciding? Get samples of $ ! US$ 15/Piece Request Sample |



Revolutionize spinal surgery with our mature implant system. This solution is designed to meet rigorous clinical standards, offering both sterile and non-sterile packaging options for maximum surgical convenience.
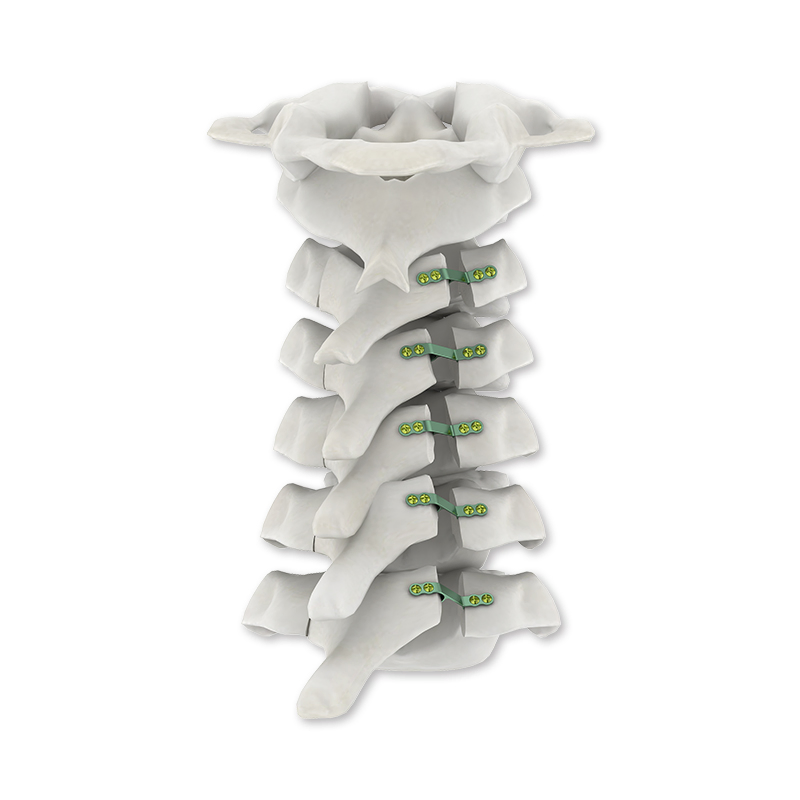
| Product Name | Spinal Internal Fixation 5.5/6.0 U-Multi-Axial Reduction Pedicle Screw |
| Material | High-Grade Titanium |
| Color | Gold, Blue, Grey, Pink, etc |
| Related Rod | 5.5 / 6.0 mm |
| Diameter | 4.5 / 5.0 / 5.5 / 6.0 / 6.5 / 7.0 mm |
| Length | 30 / 35 / 40 / 45 / 50 / 55 / 60 mm |
| Certificates | CE / ISO 13485 & 9001 |
| Package | Non-Sterilized / Sterilized Options |


Explore our Pedicle Screw System for Spinal Fusion, offering high-quality orthopedic spine instruments for professional surgical procedures.
Our pedicle screws and rods are manufactured from high-grade titanium, known for its excellent biocompatibility and mechanical strength.
Yes, we provide both sterile and non-sterile packaging to meet the specific logistics and preparation needs of different surgical facilities.
Our system is highly versatile, offering diameters ranging from 4.5mm up to 7.0mm, with lengths from 30mm to 60mm.
Yes, we offer logo printing and customization services to help enhance your brand presence in the medical market.
All our medical apparatus systems are certified under CE and ISO 13485 & 9001 standards, ensuring global compliance and safety.
We maintain an efficient production workflow that allows for quick sample delivery, often within 3 days of order confirmation.
 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants