



1 / 5










| Still deciding? Get samples of $ ! US$ 120/Piece Request Sample |


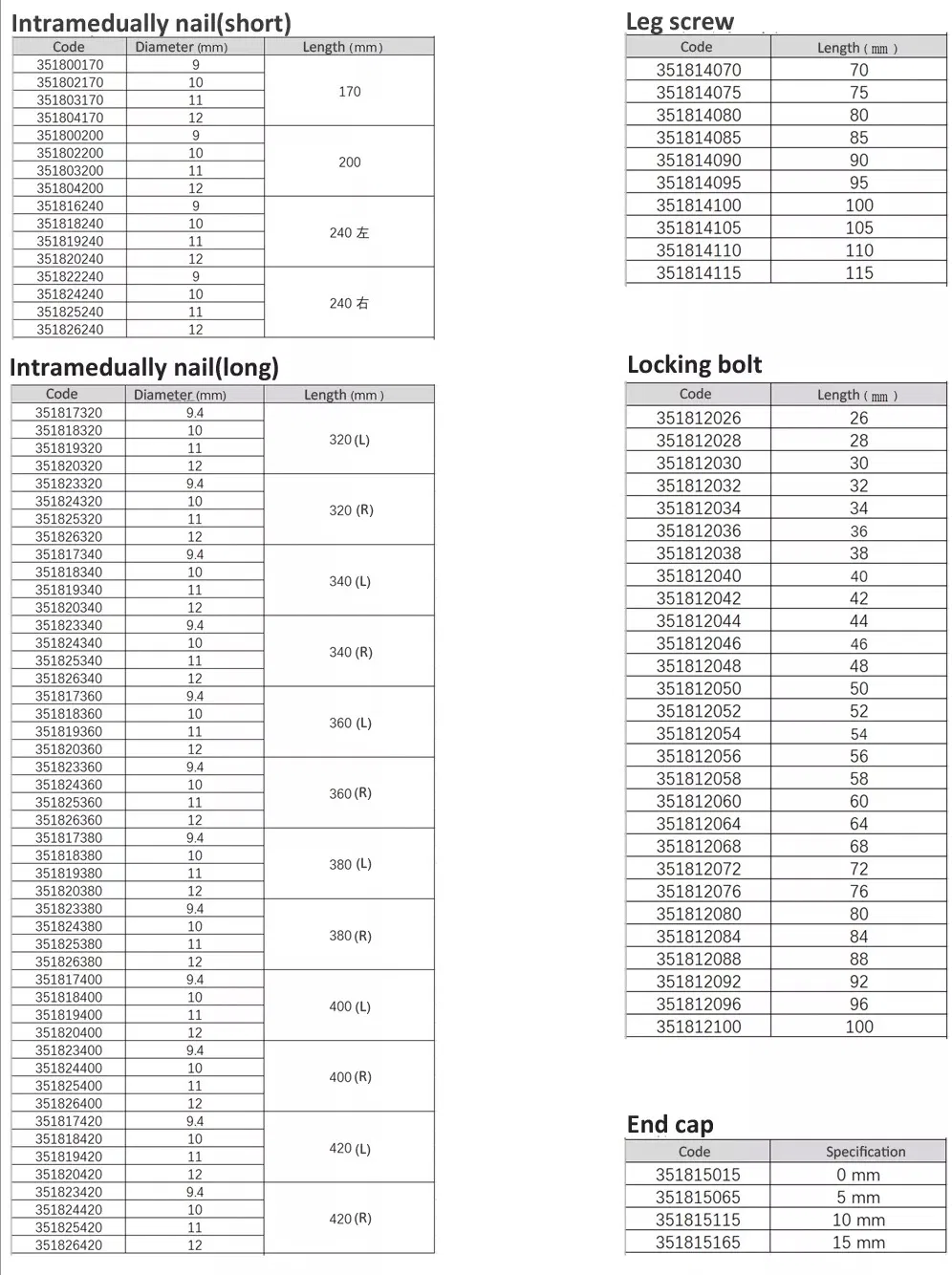
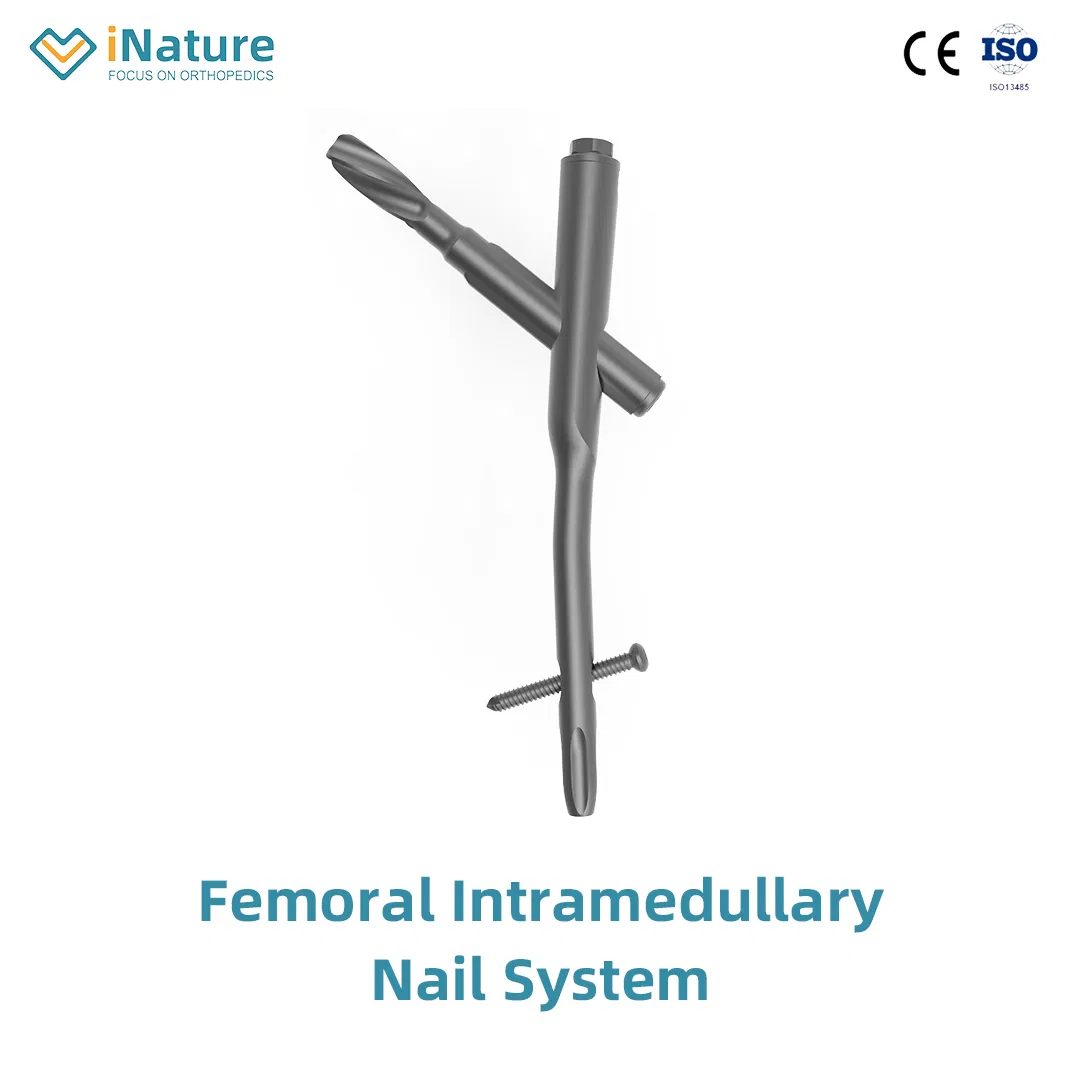
| ✓ Material: Titanium |
| ✓ Surface Finished: Oxidation / Milling for Titanium |
| ✓ Design: Medial-lateral angle of 5° for insertion at greater trochanter tip |
| ✓ Insertion: Lateral flatten design reduces stress to lateral cortical bone |
| ✓ Tip Design: Elastic groove tip reduces concentration stress |
| ✓ Safety: 240mm distal curved design prevents nail tip contact with cortical bone |
| ✓ Stability: Lag screw provides excellent compaction; ideal for osteoporotic patients |
| ✓ Efficiency: Double thread locking bolt design reduces operation time |
Standard 3 Guarantees Policy:
● One Month: Guaranteed return or exchange for quality issues.
● One Year: Free repair services for manufacturing defects.
● After One Year: Replacement parts provided at cost value.
● Technical Support: Timeless maintenance advice and remote engineering solutions.
Quality & Value: We compete not only on price but primarily on quality. Our pricing reflects the high standards of material and engineering required for medical implants, ensuring a win-win outcome for healthcare providers and patients.




 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants