

1 / 5










| Still deciding? Get samples of $ ! US$ 80/Piece Request Sample |
| Basic Information | |
|---|---|
| Model NO. | NXGGK01 |
| Sterilization | Without Ethylene Oxide Sterilization |
| Quality Guarantee | Two Years |
| Group | Adult |
| Customized | Non-Customized |
| Certification | ISO, CE |
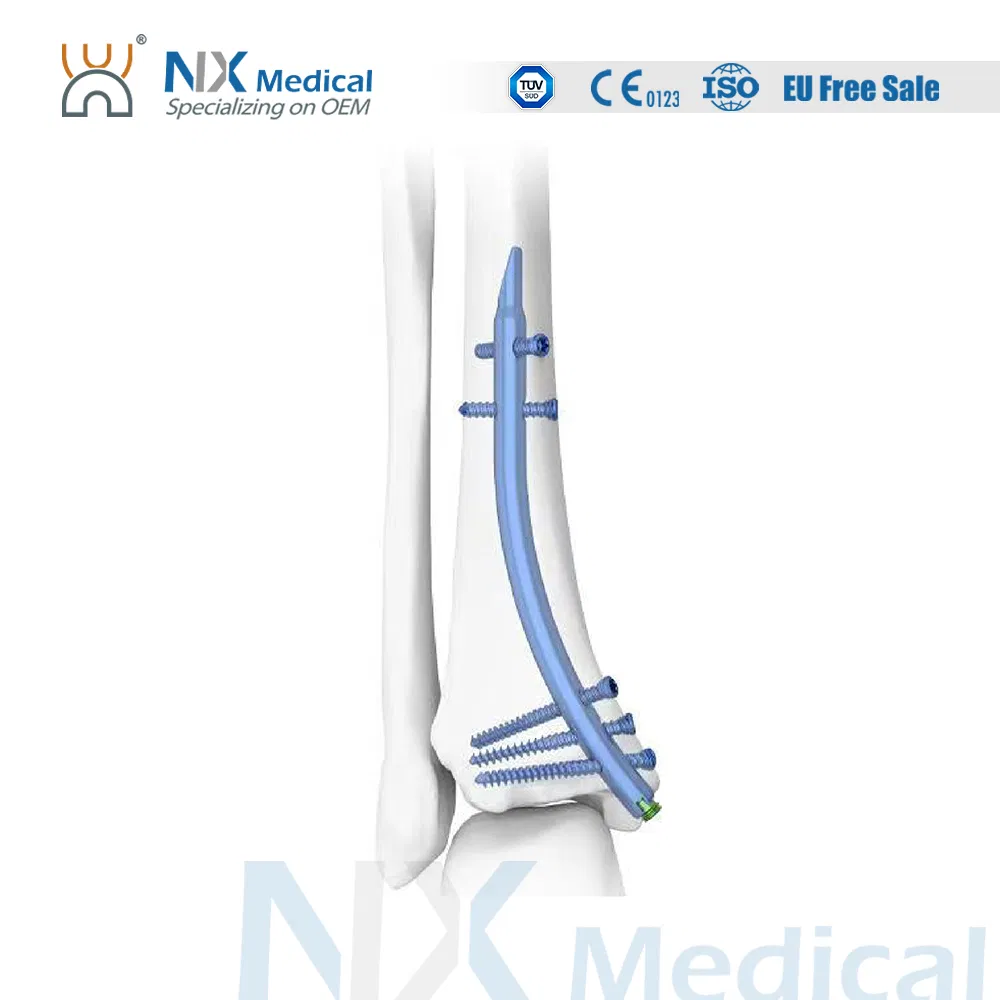
| Usage | Fracture Fixation |
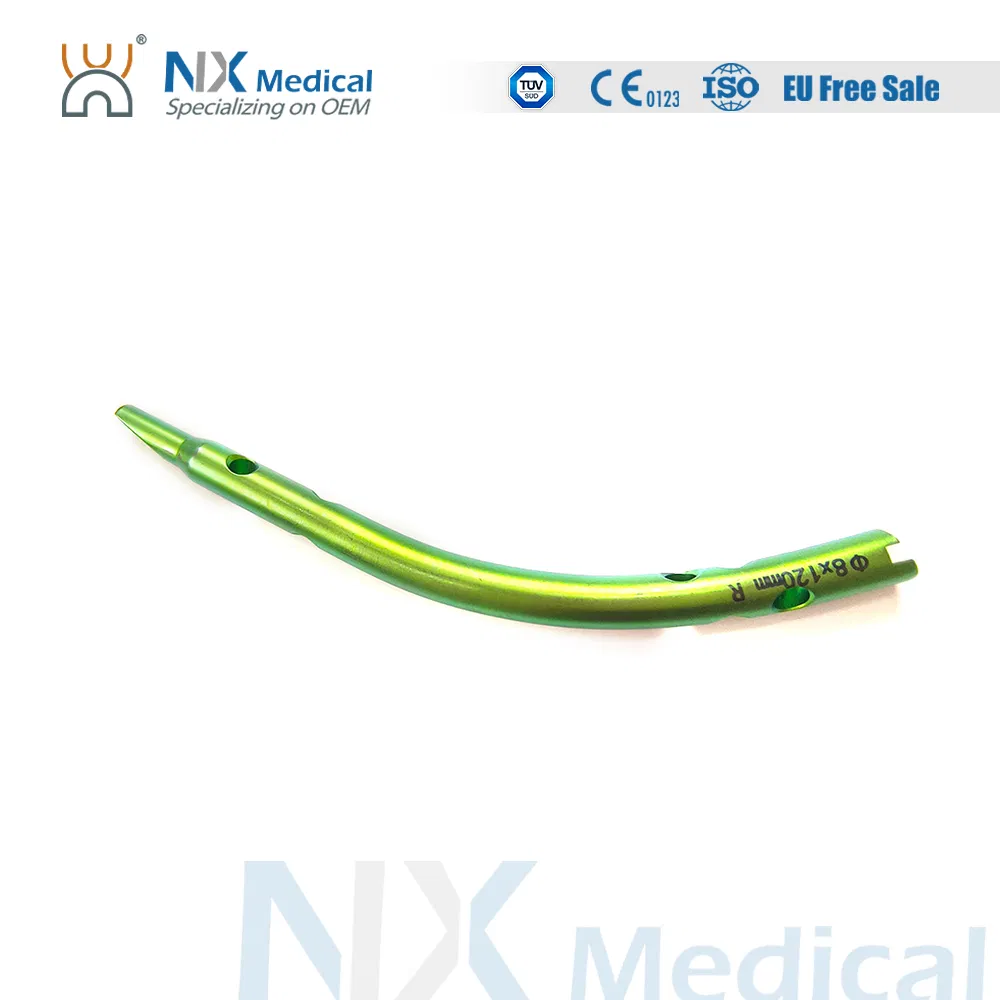
| Using Site | Tibia |
| Material | Titanium Alloy (TA) |
| Origin | China |
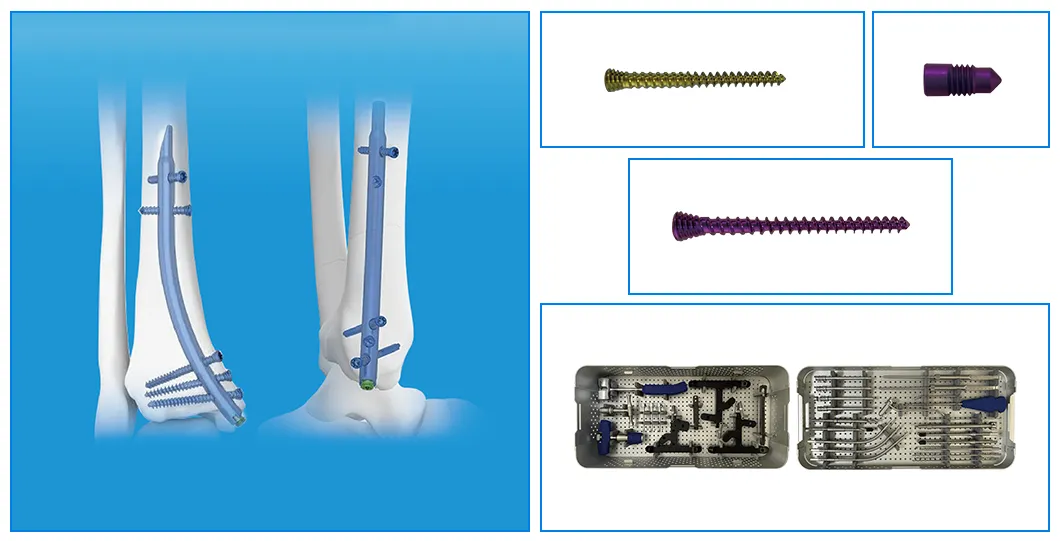
| Product | Size / Length (mm) | Material |
|---|---|---|
| Retrograde Tibial Nail | 7 x 110 / 8 x 120 / 8 x 140 | TA |
| Interlocking Cancellous Screw (φ3.5mm) | 30 to 52 (2mm increments) | TA |
| Interlocking Cortical Screw (φ3.5mm) | 18 / 20 / 22 / 24 / 26 / 28 | TA |
| Interlocking Cortical Screw (φ4.0mm) | 24 / 26 / 28 / 30 / 32 / 34 | TA |
| End Cap | Standard / 5mm | TA |


Our company is a leading manufacturer of orthopedic products, providing a comprehensive range of cost-effective surgical and reliable therapeutic solutions worldwide. We offer superior orthopedic trauma implants, spinal implants, and high-precision surgical instruments. Committed to global innovation, we continuously invest in R&D to enhance our product lines, including locking plates, intramedullary nails, and spinal fixation systems.
We operate under a strict quality management system, complying with ISO13485, with products certified by TUV CE0123 and NMPA. Our goal is to ensure the safest and most advanced implants that guarantee reliable recovery and a pain-free life for patients after surgery.




 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants