












| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |

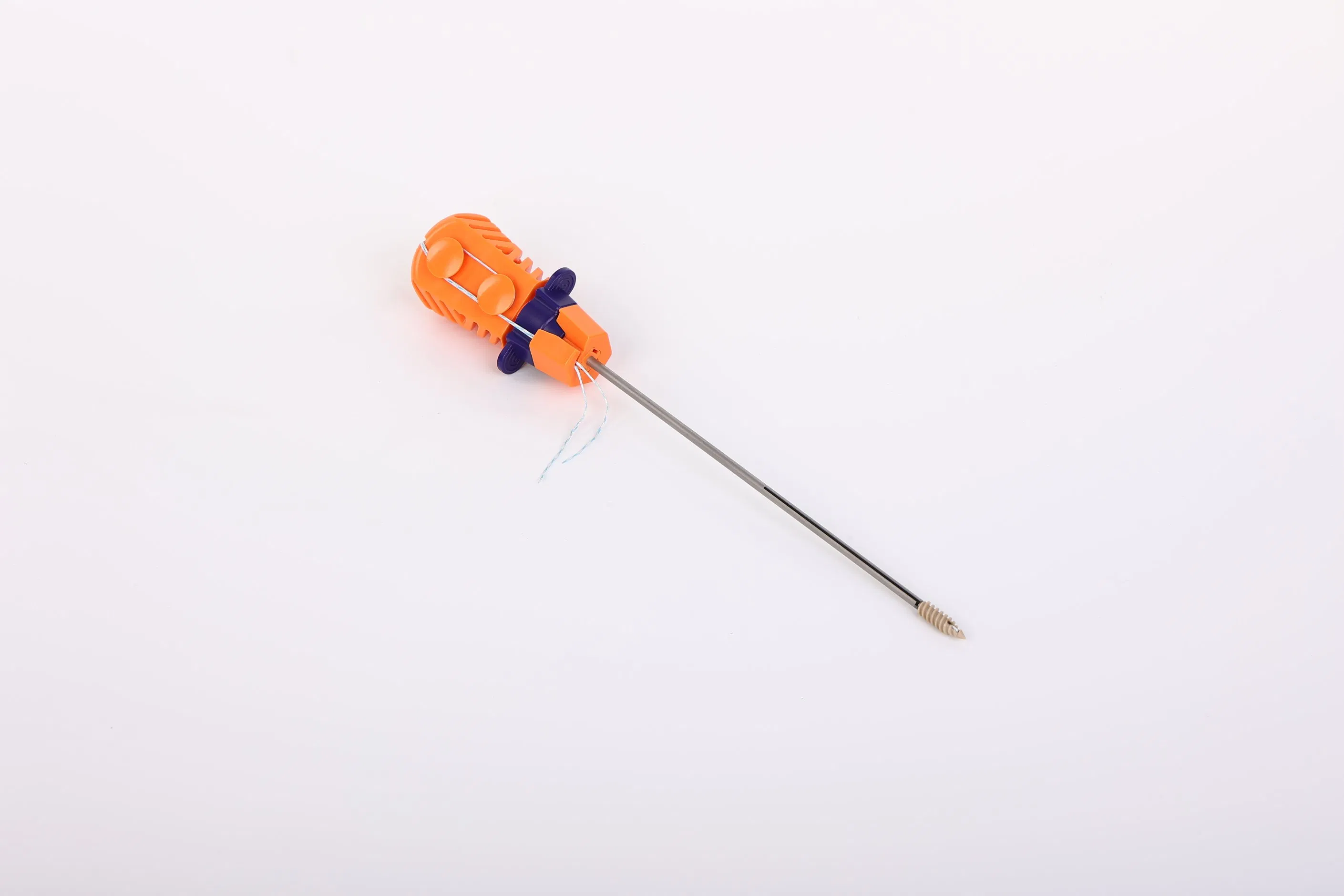
The Suture Anchor System consists of a high-performance anchor, nonabsorbable suture, and a precision inserter. The anchor is fabricated from Ti6Al4V alloy, strictly adhering to ISO 5832-3:2016 requirements. The accompanying suture is crafted from ultra-high molecular weight polyethylene (UHMWPE) material, woven for maximum strength without additional coating, meeting ASTM F2848-17 standards.

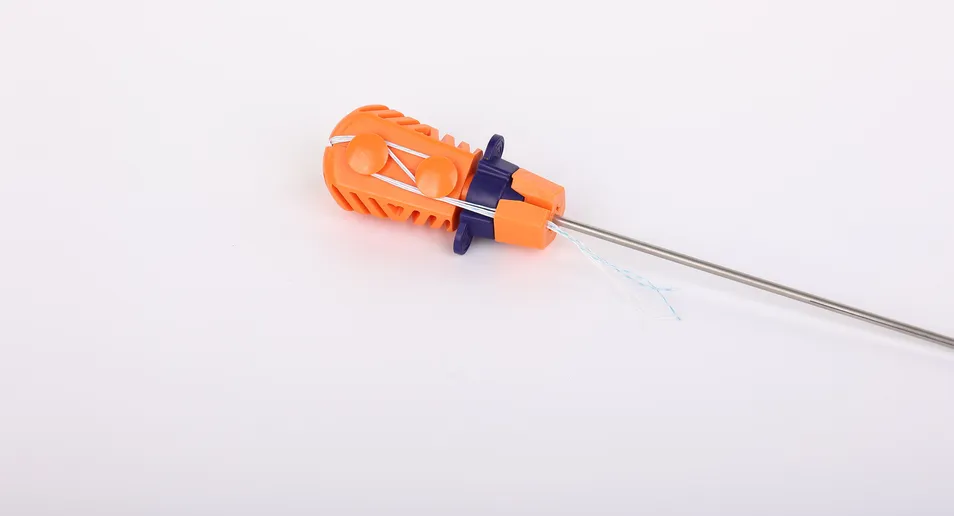
For enhanced visibility during surgery, blue sutures are available, dyed with FDA-approved materials. The inserter component that contacts the human body is manufactured from medical-grade stainless steel (ASTM F899-12b). The ergonomically designed handle, made from polycarbonate and ABS, ensures a non-slip grip for the surgeon while remaining external to the surgical site.


Our production process utilizes advanced Class 100,000 clean workshops and Class 10,000 purification laboratories to guarantee the highest safety standards. All orthopedic surgical consumables, including suture anchors and meniscal repair systems, are fully compliant with CE certification and ISO 13485 standards, ensuring reliability for medical institutions globally.


 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants