


1 / 5










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |


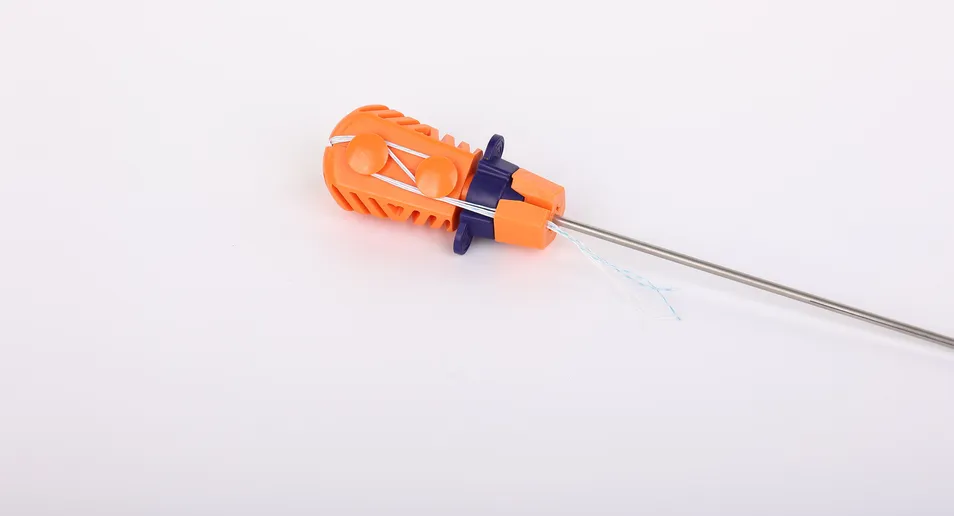

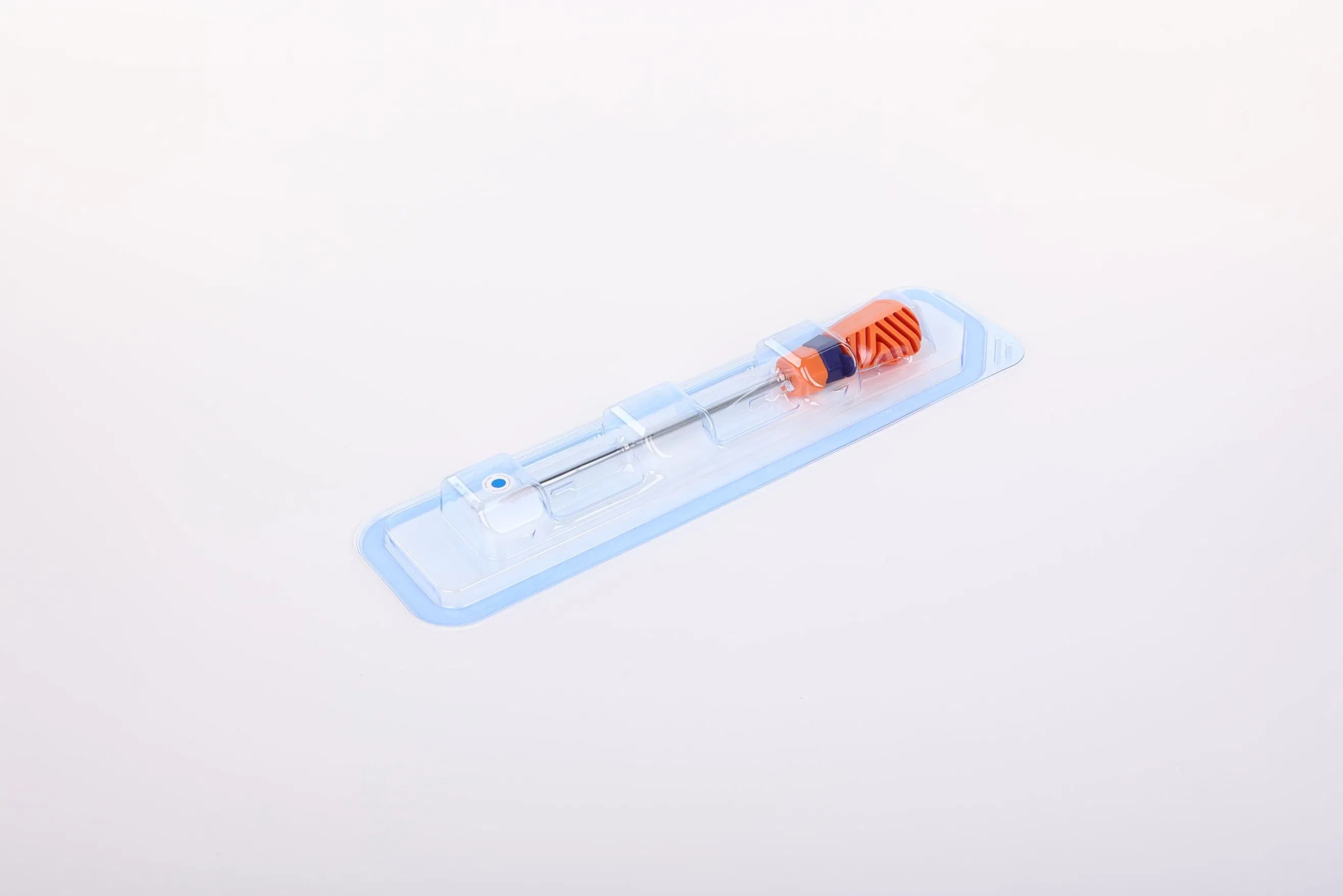
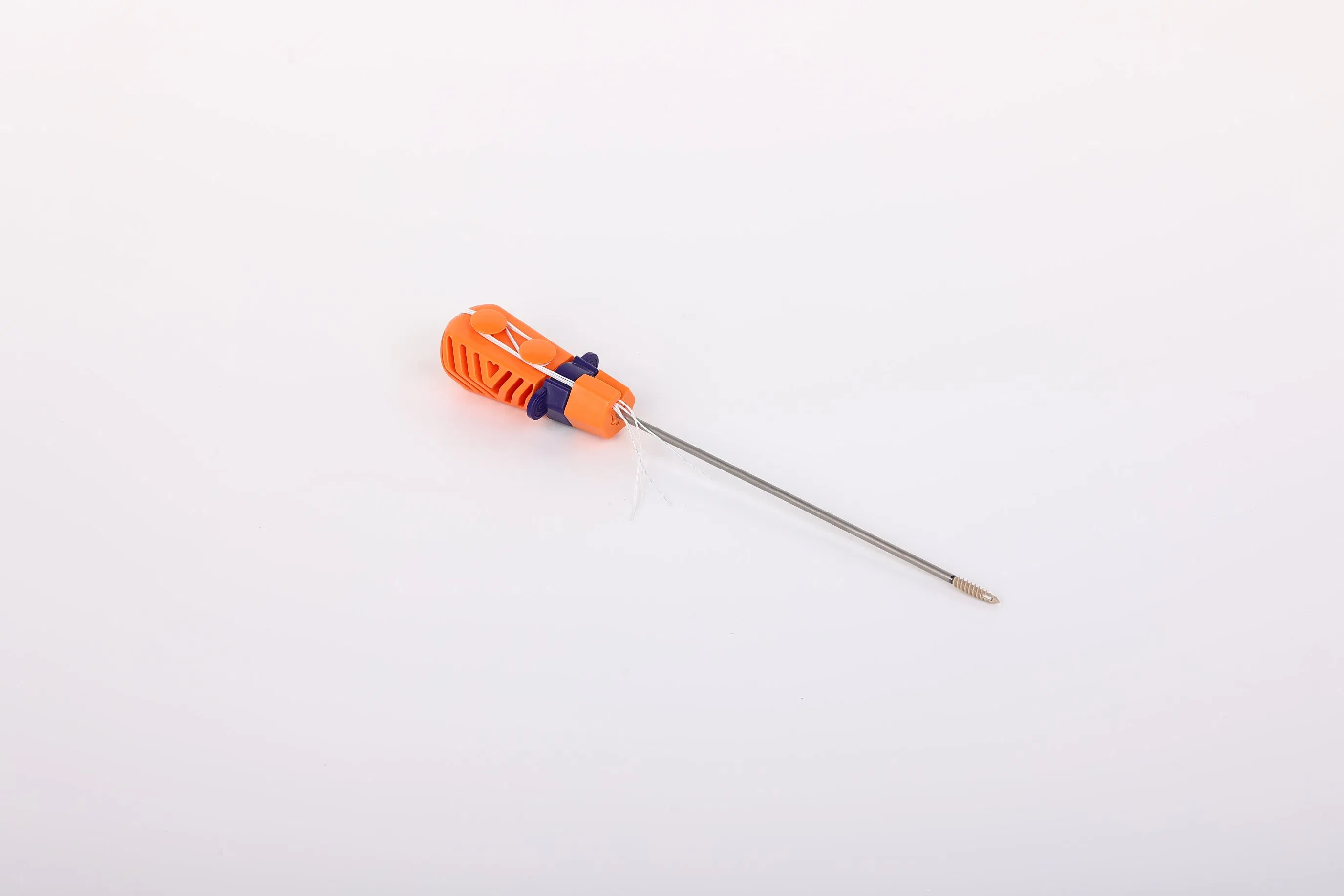
Suture Anchor System Components:



 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants