📋
Basic Information
Model NO.1500-0105-0110
MaterialTitanium
FeatureDisposable
CertificationCE, ISO13485
ColorBlue Color/Shine
SizeDetails in The Description
BrandOEM Available
UsageMaxillofacial
Specification4 holes
HS Code90211000
Production Capacity10000 PCS/Month
🔍
Product Description
| Products Name |
NO. |
Specification |
| Straight Maxillofacial Plate, Type-II |
1500-0105 |
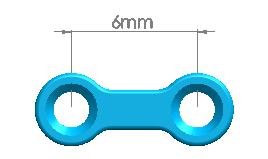
4 holes(equidistance) |
| 1500-0106 |
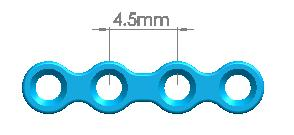
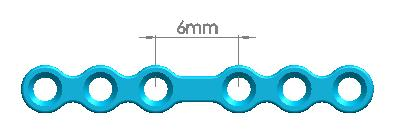
6 holes(equidistance) |
| 1500-0107 |
8 holes(equidistance) |
| 1500-0108 |
12 holes(equidistance) |
| 1500-0109 |
14 holes(equidistance) |
| 1500-0110 |
16 holes(equidistance) |
| Use: 1.5mm Self-tapping Screw & 1.5mm Self-drilling Screw |
🏢
Company Profile
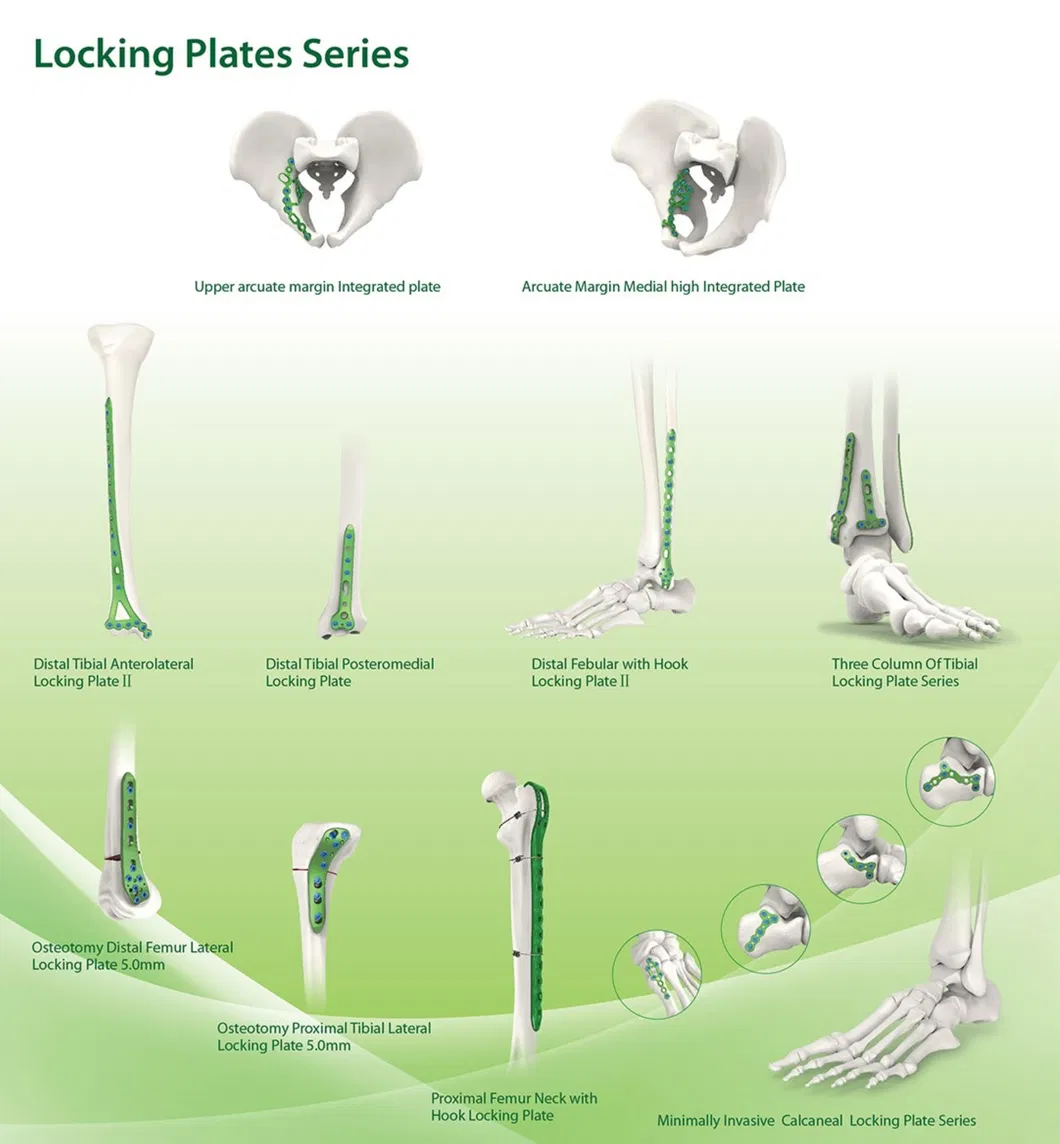
Established in 1958, the company is engaged in the development, manufacture and sales of orthopedic implants and instruments. As a key enterprise for medical apparatus, the product line covers Spine pedical screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis and surgical instruments.
All medical apparatus have passed the approval and authentication of superintendent offices. Advanced equipment, including imported CNC, high precision digital control machines and milling machines, as well as a 100,000 Grade axenic purification chamber, contribute to the high quality of the products. The manufacturer is the first to pass ISO 9001 quality system certification.
❓
Frequently Asked Questions
What is the specialization of the manufacturer?
The manufacturer specializes in orthopedic implants and instruments since 1958, including trauma plates, interlocking nails, spinal fixators, and external fixators.
How is the product quality guaranteed?
Products are produced according to EU CE standards. A pre-production sample is always provided before mass production, and a final inspection is conducted before shipment.
What categories of products can be purchased?
You can purchase Metal Bone Plates Screw Systems, Metallic Cannulated Bore Screws, Metallic Interlocking Intramedullary Nails, Spinal Fixation Devices, and External Fixation Devices.
What certifications does the production facility hold?
The facility maintains a strict quality control system with CE, ISO, and GMP certificates to ensure medical-grade precision.
What payment and delivery terms are accepted?
Accepted delivery terms include FOB, CIF, EXW, and Express Delivery. Payments can be made via T/T, L/C, Western Union, or Escrow in USD, EUR, or CNY.
Is OEM service supported for these implants?
Yes, OEM services are available for orthopedic implants and instruments to meet specific branding and specification requirements.




















 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants