The PFNA (Proximal Femoral Nail Anti-rotation) is an advanced intramedullary implant specifically designed for the treatment of hip fractures, particularly unstable intertrochanteric and proximal femoral fractures. This system achieves stable fixation by sharing the load with the bone through the intramedullary nail, significantly reducing the risk of implant loosening and failure. Made from titanium alloy, PFNA combines excellent biocompatibility with mechanical strength to ensure long-term durability and patient safety.
Core Features
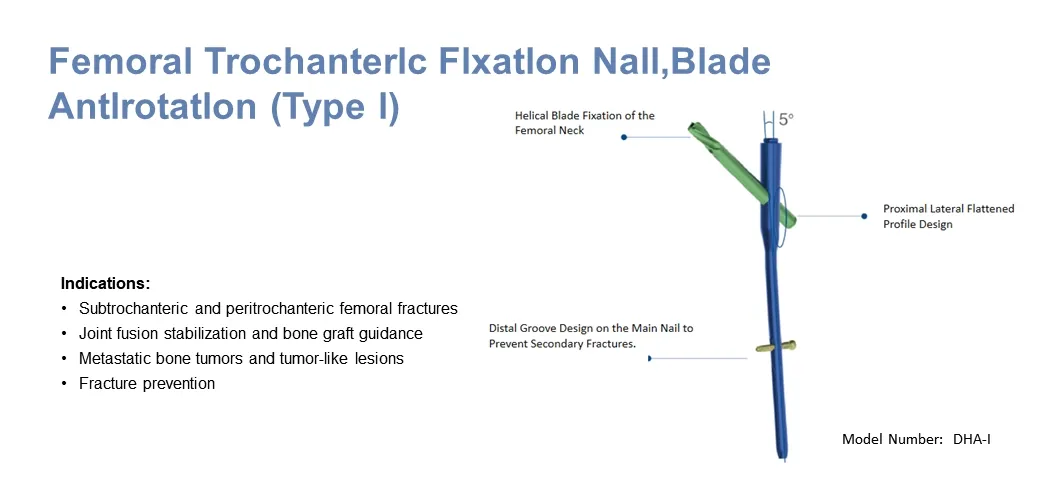
- Helical Blade Design: Compacts cancellous bone, providing rotational and angular stability, especially for osteoporotic patients.
- Anti-Rotation Mechanism: Prevents rotational movement of the nail within the femur.
- Locking System: Distal locking options provide axial stability with dynamic or static modes.
- Advanced Material: Manufactured from titanium alloy (Ti-6Al-7Nb) for strength and biocompatibility.
- Anatomical Fit: 5° outward deviation and 1500mm front arch matching human physiological structure.
- Stress Distribution: Elastic tip and groove design facilitates insertion and ideal stress distribution.
Key Advantages
- Strong Rotational Stability: Reduces cut-out risk in fracture fragments.
- Load Sharing Design: Decreases implant fatigue and fracture risk.
- Minimally Invasive: Technique reduces soft tissue damage and promotes faster recovery.
- Early Mobilization: Supports early postoperative weight-bearing and improves quality of life.
⚙
Manufacturing Excellence
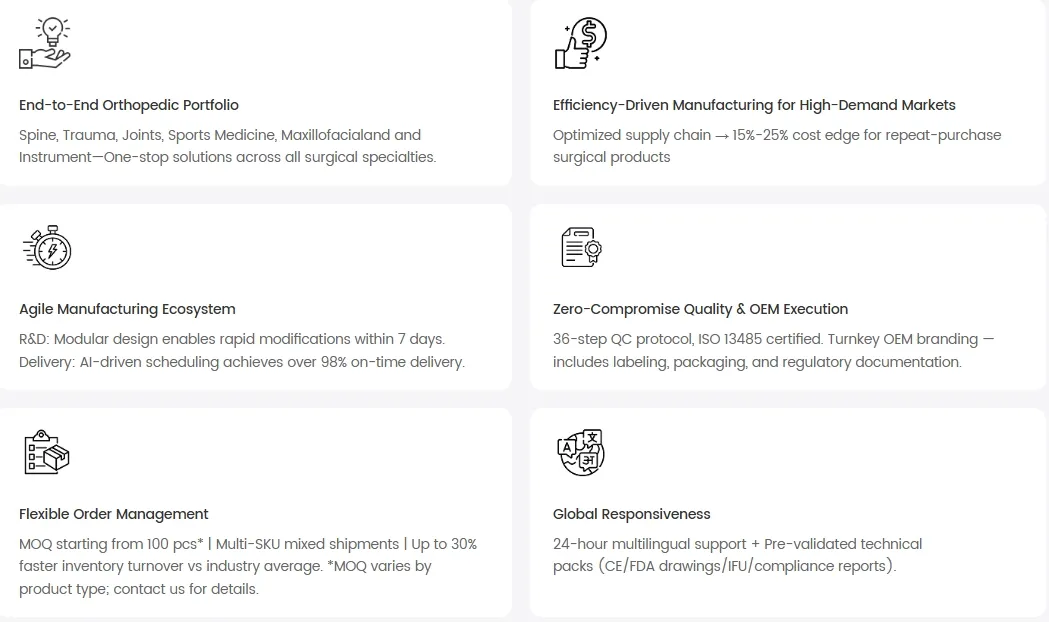
As a leading manufacturer of standardized orthopedic implants and instruments, we serve global distributors and importers with a full range covering trauma, spine, joints, sports medicine, and CMF. Our facilities include in-house R&D, certified laboratories, and modern production lines, supporting both standard supply and custom OEM/ODM solutions. We focus on reliable, scalable manufacturing to ensure quality and regulatory compliance in every medical device produced.
?
Frequently Asked Questions
Are your products certified for international medical standards?
Yes, we hold the EN ISO 13485 certification, which confirms our commitment to quality and regulatory compliance in the medical device industry, ensuring products meet stringent safety and performance requirements.
What are the shipping methods and how are costs calculated?
We typically utilize courier services or postal services upon request. Shipping costs are determined by destination, package size, and weight, provided in a detailed quote based on your specific requirements.
What is the minimum order quantity (MOQ)?
Our standard minimum order value is US$1000. We also offer sample orders at cost to allow for market testing before committing to larger volumes.
Can you manufacture custom products not listed in the catalogue?
Yes, we accommodate custom orders. By providing pictures or samples of the desired product, we can work to create a similar item. Please note that custom orders may require a longer lead time.
Do you maintain a steady stock of these implants?
We maintain stock for most products with regular monthly production cycles. However, availability varies based on demand, so we recommend consulting with the sales team for real-time inventory updates.
Why should global agents choose your orthopedic solutions?
With a track record of serving over 2,000 agents, we offer high-volume distribution capabilities, low MOQ options, and fast turnaround times suitable for both emerging and established markets.


















 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants