 Egyptian Clinical Grade Orthopedic Titanium Intramedullary PFNA Interlocking Nails
Egyptian Clinical Grade Orthopedic Titanium Intramedullary PFNA Interlocking Nails
 High-Quality Interlocking Nail Intramedullary Nail for Cairo Medical Centers
High-Quality Interlocking Nail Intramedullary Nail for Cairo Medical Centers
 Multi-Screw Hole Tibial Intramedullary Nail System - Egypt Standard
Multi-Screw Hole Tibial Intramedullary Nail System - Egypt Standard
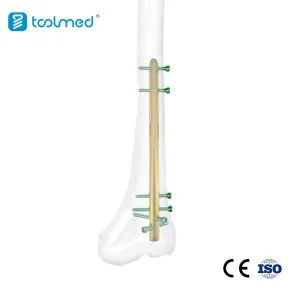 Orthopedic Trauma Interlocking Intramedullary Nail for Femur (Alexandria Surgical Spec)
Orthopedic Trauma Interlocking Intramedullary Nail for Femur (Alexandria Surgical Spec)
Egypt's healthcare sector is undergoing a profound transformation, driven by the government's "Vision 2030" and the implementation of the Universal Health Insurance System. In the realm of orthopedics, specifically Intramedullary Nails (IM Nails), the market is witnessing a surge in demand. This is primarily due to the increasing incidence of road traffic accidents (RTAs) and a growing geriatric population prone to fragility fractures in major hubs like Cairo, Giza, and Alexandria.
While Egypt has several domestic medical device distributors, the high-tech manufacturing of Grade 5 Titanium intramedullary nails remains a specialized field. Most leading hospitals prefer international partners who can provide GMP and ISO-certified implants that ensure biocompatibility and long-term stability.
The current trend in Egypt is shifting toward Minimally Invasive Surgery (MIS). Local surgeons are increasingly adopting PFNA (Proximal Femoral Nail Antirotation) systems and distal femoral nails that allow for smaller incisions and faster recovery times, reducing the burden on public healthcare facilities.
Titanium alloy (Ti6Al4V) has become the gold standard in the Egyptian market. Its superior fatigue strength and lower modulus of elasticity compared to stainless steel make it ideal for the femoral and tibial nailing systems used in complex trauma cases across the Nile Delta region.
We are a GMP and ISO certified global orthopedic implants manufacturing company, committed to designing and manufacturing products that are safe, effective, and of high quality. Our mission is to provide reliable, safe, and affordable Orthopedic implants of high geometric accuracy and surface quality to patients worldwide, aiming for a fast healing success.
For the Egyptian market, we understand the logistical and regulatory requirements set by the Egyptian Drug Authority (EDA). Our experience exporting to 75+ countries, including extensive reach in the Middle East, ensures that our intramedullary nails meet both national and international standards (CE/ISO).
Our Intramedullary Nails are designed for specific clinical scenarios common in Egyptian trauma centers:
Careful and stringent quality control is our main priority. At Shanghai XOVEC Medical Instrument Co., Ltd., every innovation undergoes complete monitoring right from its procurement to the supply.





The future of intramedullary nailing in Egypt is tied to Digital Health and Smart Implants. As Egyptian medical universities in Mansoura and Assiut push the boundaries of research, there is a growing interest in:
As a leading factory partner, XOVEC continues to invest in R&D to bring these technologies to the Egyptian medical community at an affordable price point, ensuring that "High Tech" doesn't mean "High Cost."
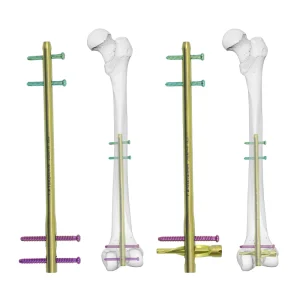 Titanium DFN Distal Femoral Nail - Cairo Hospital Grade
Titanium DFN Distal Femoral Nail - Cairo Hospital Grade
 Surgical Femoral Intramedullary Implant for Giza Trauma Centers
Surgical Femoral Intramedullary Implant for Giza Trauma Centers
 Reconstruction Femoral Nail System for Complex Egyptian Cases
Reconstruction Femoral Nail System for Complex Egyptian Cases
 Ankle Fusion Interlocking Nail - Premium Quality for Alexandria Clinics
Ankle Fusion Interlocking Nail - Premium Quality for Alexandria Clinics
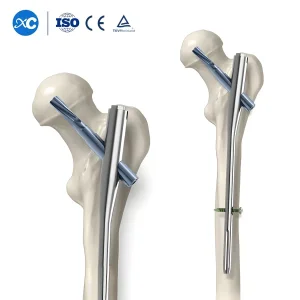 Trauma Surgery Gama Interlocking PFNA Nail - Egypt Stock
Trauma Surgery Gama Interlocking PFNA Nail - Egypt Stock
 Retrograde Intramedullary Femur Fixation 11*200 - Egyptian Standard
Retrograde Intramedullary Femur Fixation 11*200 - Egyptian Standard
 Expert Humeral Proximal Interlocking Blade Nail System
Expert Humeral Proximal Interlocking Blade Nail System
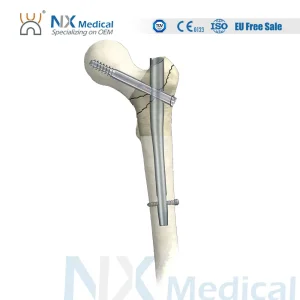 Titanium Interlocking Femoral PFNA Bone Nail - Factory Direct to Egypt
Titanium Interlocking Femoral PFNA Bone Nail - Factory Direct to Egypt