 High Quality CE ISO Certified Cruciate Ligament Circle Titanium Button-Factory Manufactured Endobutton
High Quality CE ISO Certified Cruciate Ligament Circle Titanium Button-Factory Manufactured Endobutton
 Geasure Combined External Fixator Combined Orthopedic External Fixation
Geasure Combined External Fixator Combined Orthopedic External Fixation
 China Manufacture Orthopedic Surgical Implants Laminar Hook for Posterior Cervical Fixation System Spinal Implant
China Manufacture Orthopedic Surgical Implants Laminar Hook for Posterior Cervical Fixation System Spinal Implant
 Titanium Alloy Femur Fixator for Bone Alignment
Titanium Alloy Femur Fixator for Bone Alignment
 Safe and Reliable Titanium Coating Surgical Instrument Set Orthopedic Implant Plif Lumbar Interbody Fusion Peek Cage System
Safe and Reliable Titanium Coating Surgical Instrument Set Orthopedic Implant Plif Lumbar Interbody Fusion Peek Cage System
 Smart Home Vegetable Plating Machine Indoor Hydroponics System
Smart Home Vegetable Plating Machine Indoor Hydroponics System
 Medical Surgical Instrument Orthopedic Implant Titanium Coating Banana Lumbar Peek Interbody Fusion Cage Spine System
Medical Surgical Instrument Orthopedic Implant Titanium Coating Banana Lumbar Peek Interbody Fusion Cage Spine System
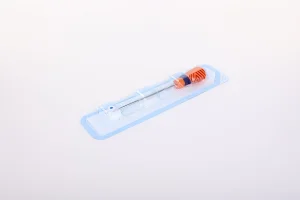 Factory Direct Price Multiple Specification Suture Anchor for Joint Repair Surgery
Factory Direct Price Multiple Specification Suture Anchor for Joint Repair Surgery
We are GMP and ISO certified global orthopedic implants manufacturing company, who remains committed to designing and manufacturing products that are safe, effective and of a high quality that meets both National and International Standards. We’re a leading orthopedic implants manufacturer and exporter based in India, with over 18+ years of experience exporting to 75+ countries.
Careful and stringent quality control is our main priority. At Shanghai XOVEC Medical Instrument Co., Ltd. every innovation undergoes complete monitoring and seamless documentation right from its procurement to the supply. Our mission is to provide reliable, safe and affordable Orthopedic implants of high geometric accuracy and surface quality, long service life and biocompatibility, to patients worldwide, aiming a fast healing success.

The global sports medicine market, particularly the segment focusing on Anterior Cruciate Ligament (ACL) and Posterior Cruciate Ligament (PCL) reconstructions, has witnessed unprecedented growth. As physical activity levels rise among the general population and professional sports become more intensive, the incidence of ligamentous knee injuries has surged. This has placed Endobuttons and various suspension fixation devices at the forefront of surgical innovation.
Modern orthopedic surgery demands implants that not only provide strong mechanical fixation but also promote faster biological integration. Suspension fixation, popularized by the Titanium Endobutton, has become a gold standard due to its high "pull-out" strength and minimal invasiveness. Globally, the shift is moving from traditional metal screws toward advanced titanium suspension systems and bioabsorbable interference screws that reduce long-term complications in patients.
Today's manufacturing of ACL/PCL fixation devices is no longer just about machining metal. It involves High-Precision CNC (Computer Numerical Control) processes that ensure tolerances are measured in microns. Materials like Grade 5 Titanium (Ti6Al4V) and PEEK (Polyether ether ketone) are meticulously selected for their biocompatibility and modulus of elasticity, which closely mimics human bone.
Furthermore, the industry is trending towards "Adjustable Loop" technology. Unlike traditional fixed-loop endobuttons, adjustable loops allow surgeons to tension the graft precisely after the button has been flipped on the femoral cortex, eliminating the need for complex pre-operative measurements and reducing graft-tunnel mismatch.

Compliance with strict regulatory frameworks ensuring legal and ethical manufacturing.

Utilizing high-grade titanium and PEEK materials with cost-efficient production models.

Deep understanding of international logistics, customs, and medical import regulations.

OEM/ODM services to meet specific clinical needs or regional surgical preferences.
For international distributors and hospital procurement departments, the selection of an ACL/PCL Fixation supplier hinges on three pillars: Traceability, Certification, and Scalability. As an established manufacturer, Shanghai XOVEC ensures every Endobutton and interference screw can be traced back to its raw material heat lot. Our facilities are equipped to handle large-scale tenders from national health ministries while maintaining the agility to support private clinical groups.
In regions like Europe and North America, the demand is high for CE-marked titanium buttons that offer superior radiological transparency. In emerging markets like Southeast Asia and Latin America, the focus is on achieving high-tier clinical outcomes with cost-effective implants that allow for broader patient access to advanced orthopedic surgeries.
Choosing Shanghai XOVEC as your Chinese manufacturing partner offers distinct advantages in the current global economic landscape:
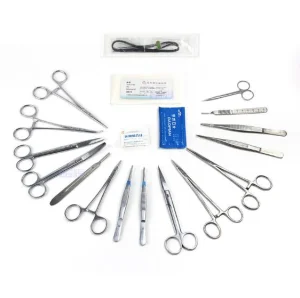 Medical Hospital Operating Room Surgical Orthopedic Instrument Set
Medical Hospital Operating Room Surgical Orthopedic Instrument Set
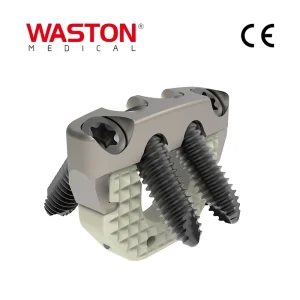 Disposable Surgical Neulen Medical Products Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Implant Zero-X Cage System
Disposable Surgical Neulen Medical Products Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Implant Zero-X Cage System
 Hip Joint System Instrument Sets with Cups for Orthopedic Surgery Surgical Instrument
Hip Joint System Instrument Sets with Cups for Orthopedic Surgery Surgical Instrument
 Orthopedic Products Supplier Acl Cannulated Interference Screw
Orthopedic Products Supplier Acl Cannulated Interference Screw
 Orthopedic External Fixator - Ilizarov External Fixator- C Ring (Stainless Steel)
Orthopedic External Fixator - Ilizarov External Fixator- C Ring (Stainless Steel)
 Titanium Alloy Anatomical Hook Guide Pin for Anterior Cervical Plate Fixation Surgery
Titanium Alloy Anatomical Hook Guide Pin for Anterior Cervical Plate Fixation Surgery
 Orthopedic Medical Equipment Retrograde Tibia Nail Instrument Set Surgical Medical Equipment
Orthopedic Medical Equipment Retrograde Tibia Nail Instrument Set Surgical Medical Equipment
 Insulation High Temperature Resistance Plastic Hexagon Allen Socket Hex Bolt Round Pan Head Peek Screws
Insulation High Temperature Resistance Plastic Hexagon Allen Socket Hex Bolt Round Pan Head Peek Screws