 Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
 S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
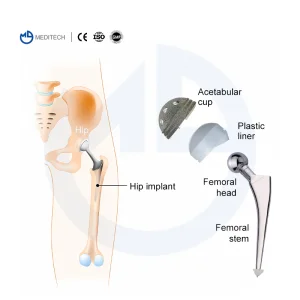 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
 Total Hip Replacement Prosthesis Corail Femoral Stem, Artificial Hip Joint System
Total Hip Replacement Prosthesis Corail Femoral Stem, Artificial Hip Joint System
 Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
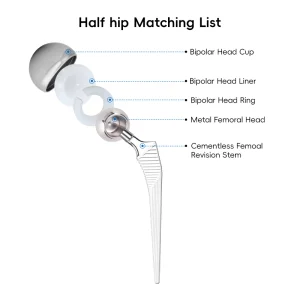 Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Instrument Set
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Instrument Set
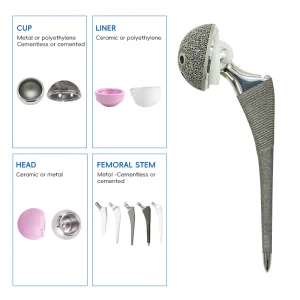 UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System
UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System
 Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
In the modern era of orthopedics, Cementless Hip Stems have become the gold standard for primary total hip arthroplasty (THA), particularly among younger, active patients. Unlike traditional cemented methods, cementless technology relies on osseointegration—the direct structural and functional connection between living bone and the surface of a load-bearing implant.
Globally, the market is witnessing a CAGR of over 5.5%, driven by the clinical success of tapered wedge designs and hydroxyapatite (HA) coatings. Manufacturers in China have transitioned from simple fabrication to complex R&D, providing high-friction surfaces and anatomically optimized geometries that rival top-tier European and American brands.
Additive manufacturing allows for the creation of "Trabecular Metal" structures that mimic the architecture of human bone, drastically improving the speed of stabilization.
Modern stems are now designed with digital twin technology, compatible with robotic systems like Mako or ROSA for sub-millimeter placement accuracy.
Bone-preserving "Short Stems" are gaining traction in Europe and Asia, allowing for easier revision surgeries later in a patient's life.
Strategic sourcing for Hospitals, Distributors, and Medical Tenders.
Professional buyers today are not just looking for a product; they are looking for a compliant supply chain. Whether you are a distributor in Latin America or a private hospital group in Southeast Asia, procurement needs revolve around three pillars:
Located in the heart of China's medical industrial hub, Shanghai XOVEC Medical Instrument Co., Ltd. leverages state-of-the-art CNC machining and vacuum plasma spraying technologies. Our advantage lies in our ability to produce premium Ti6Al4V alloy implants at a cost-to-quality ratio that is unattainable in Western markets.
We combine German-grade materials (such as UHMWPE inserts) with Chinese manufacturing agility, ensuring that high-performance hip systems remain affordable for public healthcare systems worldwide without compromising on patient safety.

We are GMP and ISO certified global orthopedic implants manufacturing company, committed to safety and geometric accuracy.




Every innovation undergoes complete monitoring and seamless documentation right from its procurement to the supply. Our mission is to provide reliable, safe and affordable Orthopedic implants of high geometric accuracy and surface quality, long service life and biocompatibility, to patients worldwide, aiming a fast healing success.
 German UHMWPE Insert Acetabular Cup Hip Joint System
German UHMWPE Insert Acetabular Cup Hip Joint System
 Replacement Constrained Liners Total Hip Arthroplasty Lining
Replacement Constrained Liners Total Hip Arthroplasty Lining
 China Factory OEM Support Thr/Tkr Replacement Instrument Set
China Factory OEM Support Thr/Tkr Replacement Instrument Set
 Veterinary Surgical Hip Joint Total Replacement Instrument
Veterinary Surgical Hip Joint Total Replacement Instrument
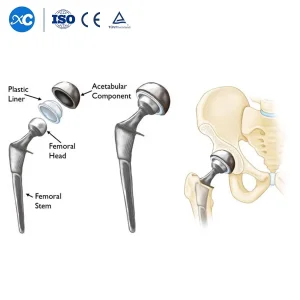 Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
 Nx Medical Orthopedic Variable Angle Pediatric Locking Plate
Nx Medical Orthopedic Variable Angle Pediatric Locking Plate
 Best Performance Surgical Power Drill Total Hip Replacement
Best Performance Surgical Power Drill Total Hip Replacement
 Total Hip Replacement for Osteoarthritis Secondary to Hip Dysplasia
Total Hip Replacement for Osteoarthritis Secondary to Hip Dysplasia
At the core of a successful Cementless Hip Stem is the material science. We utilize Ti-6Al-4V ELI (Extra Low Interstitial) titanium alloy, which offers the perfect balance of fatigue strength and modulus of elasticity closer to that of human cortical bone. This helps in reducing "stress shielding"—a common complication where the implant carries too much load, causing the surrounding bone to weaken.
Our manufacturing process includes Grit Blasting and Vacuum Plasma Spraying (VPS) to create a micro-porous surface. This roughness increases the initial primary stability through "press-fit" fixation. To further enhance long-term secondary stability, our stems often feature a Hydroxyapatite (HA) coating. HA is a bioactive material that encourages bone cells (osteoblasts) to grow directly onto the implant, creating a biological bond that can last decades.
Understanding local clinical needs is vital. In the European market, there is a high demand for Corail-style tapered stems that allow for minimally invasive surgery (MIS) approaches. In Asian markets, where developmental dysplasia of the hip (DDH) is more prevalent, we provide specialized S-ROM modular stems that allow surgeons to adjust the version and height of the femoral neck independently of the distal stem fixation.
As an exporter with over 18 years of history, our quality control protocols involve 100% inspection of critical dimensions using automated CMM (Coordinate Measuring Machines). We understand that every implant we export represents a patient's mobility and quality of life. By choosing a manufacturer with extensive E-E-A-T credentials, you ensure that your medical supply chain is robust, safe, and scientifically advanced.