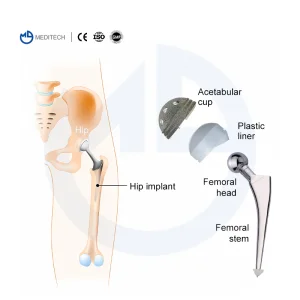
 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
 Medical Well-Leg Positioners Knee Platform Positioner / Total Knee Replacement / Tkr Positioner
Medical Well-Leg Positioners Knee Platform Positioner / Total Knee Replacement / Tkr Positioner
 Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement
Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement
 OEM Orthopedic Surgical Instrument Femoral Condyle Replacement Artificial Knee Joint Implants
OEM Orthopedic Surgical Instrument Femoral Condyle Replacement Artificial Knee Joint Implants
 OEM Orthopedic Surgical Instrument Femoral Condyle Replacement Artificial Knee Joint Implants
OEM Orthopedic Surgical Instrument Femoral Condyle Replacement Artificial Knee Joint Implants
 Tka/Tkr Primary Total Knee Joint Prosthesis Knee Orthopedic Implants Artificial Knee Replacement
Tka/Tkr Primary Total Knee Joint Prosthesis Knee Orthopedic Implants Artificial Knee Replacement
 Knee Surgery Holder, Knee Arthroscopic Surgery Support/ Total Knee Replacement Surgery Support/Thigh Support Knee Surgery
Knee Surgery Holder, Knee Arthroscopic Surgery Support/ Total Knee Replacement Surgery Support/Thigh Support Knee Surgery
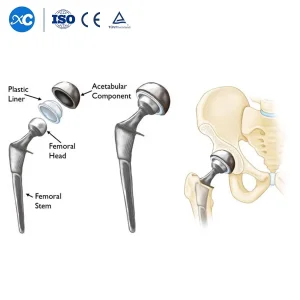 Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
Artificial Disc Replacement (ADR), also known as Total Disc Replacement (TDR), has emerged as a revolutionary alternative to traditional spinal fusion. While spinal fusion aims to eliminate pain by stopping motion in a vertebral segment, ADR focuses on motion preservation. As of 2024, the global spinal implants market is witnessing a significant shift towards non-fusion technologies, with the ADR segment projected to reach a valuation of over USD 2.5 billion by 2030, growing at a CAGR of 11.4%.
The orthopedic industry is no longer just about titanium and polyethylene. We are entering the era of 3D-printed porous structures that mimic bone trabeculae, fostering better osseointegration. As a leading China Artificial Disc Replacement supplier, we integrate AI-driven design protocols to customize the biomechanical properties of our implants, ensuring they match the physiological load of different patient demographics. Key trends include the use of PEEK (Polyetheretherketone) for radiolucency and the development of cervical and lumbar discs that offer six degrees of freedom.
Our facilities utilize multi-axis CNC machining and laser-sintering technologies. This allows us to maintain tolerances within microns, essential for the articulating surfaces of artificial discs to prevent wear and debris-induced osteolysis.
Located in the heart of Shanghai's medical hub, we leverage a robust ecosystem of raw material providers (Titanium Gr 5, UHMWPE) and logistics networks, ensuring shorter lead times for high-volume global procurement.
By optimizing the production cycle through AI monitoring, we provide premium-grade orthopedic solutions at a competitive price point, enabling hospitals worldwide to offer affordable life-changing surgeries.
Expertise Note: Modern Artificial Disc Replacement involves complex mechanics. A reliable supplier must understand the "Instantaneous Axis of Rotation" (IAR). Our R&D team ensures that every disc we manufacture replicates the natural biomechanics of the human spine, reducing adjacent level degeneration—a common failure in traditional fusion.
We are a GMP and ISO certified global orthopedic implants manufacturing company. We remain committed to designing and manufacturing products that are safe, effective, and of a high quality that meets both National and International Standards. As a leading orthopedic implants manufacturer and exporter, we combine 18+ years of experience with a footprint in over 75+ countries.

Careful and stringent quality control is our main priority. At Shanghai XOVEC Medical Instrument Co., Ltd. every innovation undergoes complete monitoring and seamless documentation right from its procurement to the supply. Our mission is to provide reliable, safe, and affordable Orthopedic implants of high geometric accuracy and surface quality, long service life and biocompatibility, to patients worldwide, aiming a fast healing success.




The demand for Artificial Disc Replacement varies by region, but the core requirement remains consistent: biocompatibility and durability. In aging populations like those in Europe and North America, ADR is used to treat Degenerative Disc Disease (DDD) in active seniors who wish to maintain their lifestyle. In emerging markets, the demand is driven by a younger workforce suffering from cervical issues due to prolonged sedentary desk work.
For international medical device distributors, sourcing from a reliable China factory offers the flexibility of OEM and private labeling. We provide comprehensive documentation support for local regulatory registrations (FDA, CE, SFDA), ensuring that our partners can enter their local markets with confidence. Our customized manufacturing capability means we can adapt disc heights and footprints to suit specific regional anatomical variations.
We utilize AI-based finite element analysis (FEA) to simulate 10 million cycles of movement for our artificial discs. This "digital twin" testing ensures that every batch of products exceeds the safety requirements for long-term implantation, reducing the risk of revision surgery.
 Total Hip Replacement Prosthesis Corail Femoral Stem, Artificial Hip Joint System Titanium Hip Joint Prosthesis Implant
Total Hip Replacement Prosthesis Corail Femoral Stem, Artificial Hip Joint System Titanium Hip Joint Prosthesis Implant
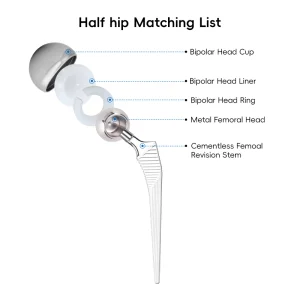 Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set
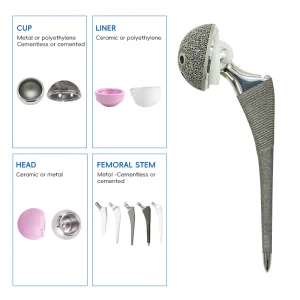 UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System Total Knee Replacement Prosthesis Implants Instruments Set
UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System Total Knee Replacement Prosthesis Implants Instruments Set
 Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
 Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
 China Factory OEM Support Thr/Tkr Total Knee and Hip Joint Replacement Instrument Set
China Factory OEM Support Thr/Tkr Total Knee and Hip Joint Replacement Instrument Set
 Total Knee Replacement Internal Stitches Surgical Suture
Total Knee Replacement Internal Stitches Surgical Suture
 Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
Choosing the right Artificial Disc Replacement supplier is a decision that impacts patient outcomes for decades. At Shanghai XOVEC, we combine the experience of an 18-year industry veteran with the innovation of a technology-driven Chinese factory. Our commitment to motion preservation, mechanical integrity, and surgical precision makes us the preferred partner for orthopedic surgeons and distributors worldwide. Whether you are looking for standardized cervical discs or custom lumbar solutions, our team is equipped to deliver excellence that meets the highest E-E-A-T standards in the medical field.