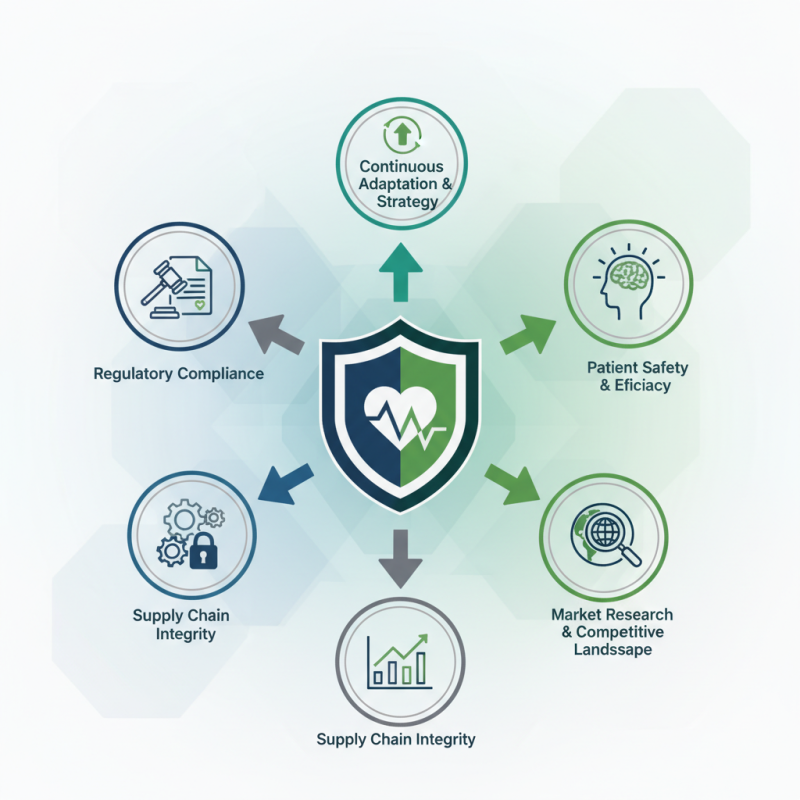
Importing orthopedic implants involves navigating a complex landscape of regulations, quality control, and market demand. As Dr. Emily Chen, a leading expert in orthopedic materials, observes, "Understanding market needs is crucial for successful imports." With the heightened focus on patient safety and implant efficacy, key considerations emerge.
What are the key points of importing orthopedic implants? This question deserves attention. First, regulatory compliance is essential. Each market has unique regulations. These rules ensure that implants meet safety standards. Failing to comply can lead to serious consequences.
Another critical point is supply chain integrity. Quality management must be maintained throughout the process. Any lapse can jeopardize patient health. Additionally, it's vital to understand the competitive landscape. Importing without market research can be a costly mistake. In this ever-evolving field, adapting strategies based on insights is not just beneficial; it's necessary. Balancing all these aspects is challenging. Continuous reflection and adjustment are required to sustain success in this industry.
Importing orthopedic implants is a complex process that requires a solid understanding of regulatory standards. Many countries have stringent guidelines to ensure the safety and efficacy of these medical devices. For instance, the Global Harmonization Task Force (GHTF) emphasizes the need for compliance with risk management and clinical evaluation during the importation process.
Regulatory standards often vary by region. In the United States, the Food and Drug Administration (FDA) places implants into classifications based on risk. Class III devices, which include many orthopedic implants, require premarket approval. According to a 2021 market study, around 75% of orthopedic implant manufacturers cite regulatory compliance as a significant hurdle in their operations. This highlights the importance of staying informed about changing regulations.
Tips: Always consult professionals with experience in orthopedic regulations. Regularly review updates from regulatory bodies to ensure compliance. Consider using third-party services for additional guidance. Regularly auditing your import processes can help identify potential gaps in adherence to regulations, allowing for timely adjustments. Keeping an eye on global trends in orthopedic device approvals can also enhance your competitive positioning.
When importing orthopedic implants, finding reliable suppliers is essential. Start by researching manufacturers with proven track records. Look for those with quality certifications. Check if they have experience in orthopedic products. Visit their facilities if possible. This helps you grasp their production capabilities.
Additionally, consider supplier references. Communicate with other healthcare providers who have worked with them. This can reveal insights into their reliability. Evaluate customer service as well. A responsive supplier can streamline your import process. Poor communication may lead to delays and misunderstandings.
Don't overlook regulatory compliance. Make sure the suppliers follow relevant medical standards. However, this can be challenging. Some manufacturers may provide counterfeit certifications. Always verify authenticity. Be prepared to ask tough questions and seek transparency in their processes. This diligence can save you potential issues down the line.
Importing orthopedic implants is a critical process that demands strict quality control. Assessing quality starts with understanding the product standards. Ensuring that implants meet international safety regulations is essential. Non-compliance can lead to serious consequences for patients and healthcare providers.
Quality control should not be taken lightly. It involves rigorous testing and validation processes. Manufacturers must provide detailed documentation. This includes certificates of compliance and test results. The risk of importing substandard products remains high. Transparency in the supply chain is vital. Inadequate information can lead to distrust and safety issues.
Compliance checks should be part of routine assessments. Regular audits of suppliers ensure they meet required standards. However, many importers overlook this step. They may aim for cost savings rather than quality assurance. This choice can compromise patient safety. Continuous education about regulations and standards is necessary for all stakeholders.
When importing orthopedic implants, understanding tariffs and customs regulations is essential. The medical device industry faces complex rules. According to a 2022 report, inconsistencies can lead to delays and increased costs. It's crucial for importers to stay updated on these regulations.
Tariffs on medical devices can vary significantly. For instance, in 2021, the average tariff rate was around 3.5%, depending on the product classification. However, some specific categories may face much higher rates. This unpredictability makes thorough research vital. Experienced importers often advise using the Harmonized System (HS) codes to ensure proper classification.
Customs regulations also require careful navigation. Importers must provide accurate documentation, including bills of lading and certificates of origin. Inaccuracies can trigger audits or customs holds. Some importers learn this the hard way. Compliance with both local and international regulations is non-negotiable. Understanding this landscape better can lead to smoother importing processes and enhanced business operations.
When planning logistics for importing orthopedic implants, efficiency is crucial. A solid strategy ensures timely delivery and minimizes costs. Collaborating with experienced freight forwarders can streamline this process. They understand customs regulations and can offer valuable insights.
Tips: Consider using software for real-time tracking. This adds transparency to your supply chain. You may encounter delays due to unforeseen circumstances. Having contingency plans in place is essential.
Distribution strategies must also be tailored to your target market. Knowing where to send your products is key. Partner with healthcare providers who have established networks. This facilitates easier access to your implants.
Tips: Keep communication open with your partners. Misunderstandings can lead to significant setbacks. Regularly review your strategies to adapt to market changes. Flexibility is important in a rapidly evolving environment.
: Research manufacturers with proven track records and quality certifications.
Look for experience in orthopedic products and check references from other healthcare providers.
It ensures compliance with international safety regulations and protects patient safety.
Providers should offer certificates of compliance and testing results for their products.
Stay updated on tariff rates and use Harmonized System (HS) codes for proper classification.
Poor communication can lead to delays and misunderstandings, complicating the import process.
Yes, regular audits help ensure suppliers meet the required standards and maintain safety.
Always verify authenticity. Ask direct questions about their processes.
Non-compliance can lead to serious consequences for patients and healthcare providers.
Understand tariffs, customs regulations, and maintain transparency with suppliers.
When considering what are the key points of importing orthopedic implants, it is essential to begin with an understanding of regulatory standards that govern the importation process. Compliance with these regulations ensures that the implants meet safety and efficacy criteria. Identifying reliable suppliers and manufacturers is crucial, as quality assurance is directly linked to the reputation and trustworthiness of the source.
Moreover, assessing quality control and compliance throughout the import process is vital to maintaining high standards. Navigating tariffs and customs regulations tailored for medical devices can significantly impact costs and logistics. Lastly, effective planning of logistics and distribution strategies is necessary for timely delivery and availability of the implants in the target market. Bringing these elements together ensures a successful importation process for orthopedic implants.

 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants