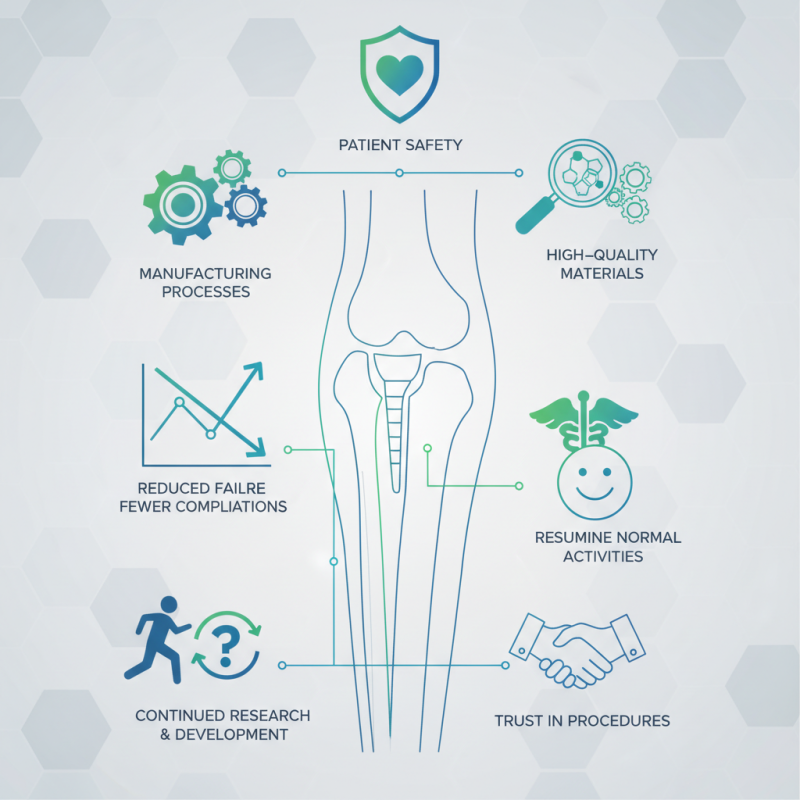
Quality management plays a crucial role in the development and success of orthopedic implants. An expert in this field, Dr. Emily Carter, emphasizes, "Quality management ensures that every implant meets the highest standards for patient safety." This statement highlights the significance of adhering to stringent quality protocols to prevent complications.
Orthopedic implants must function reliably over time. Quality management innovations can drastically improve manufacturing processes. Ensuring high-quality materials and precise engineering reduces failure rates significantly. Many hospitals report fewer complications linked to better-monitored implants.
Patients rely on these devices for resuming normal activities. Poor quality can lead to serious issues, including patient dissatisfaction and costly revisions. Therefore, addressing quality management is essential for maintaining trust in orthopedic procedures. The question remains, "What is the role of quality management in orthopedic implants?" This inquiry drives continued research and development in the industry.
Quality management systems (QMS) play a crucial role in the production of orthopedic implants. These systems ensure that products meet necessary safety and efficacy standards. According to a report from the International Organization for Standardization (ISO), effective QMS can reduce defects in manufactured products by 20% to 50%. This reduction minimizes complications for patients and leads to better surgical outcomes.
Many orthopedic manufacturers face challenges in implementing these systems. The complexities in the design and materials used in implants can complicate quality control. Data shows that 30% of companies in the orthopedic sector reported issues related to compliance with quality standards. Streamlining processes and investing in training are vital steps in overcoming these hurdles.
Tips: Regular audits can reveal potential weaknesses in a QMS. Fostering a culture of continuous improvement is essential. Engaging all employees in quality training creates a more robust QMS. Such practices can lead to fewer recalls and higher patient satisfaction. Ultimately, a solid quality management framework is not just a regulatory requirement; it is a commitment to patient safety and care.
Quality management for orthopedic implants involves strict standards and regulations. These guidelines ensure that products are safe and effective for patients. The International Organization for Standardization (ISO) has established specific standards, such as ISO 13485, focused on quality management systems. This standard emphasizes the need for manufacturers to maintain rigorous documentation and process controls.
Compliance with regulatory bodies, like the FDA in the United States, is essential. The FDA mandates premarket approval for many orthopedic devices. Manufacturers must submit extensive clinical data to prove the safety and effectiveness of their products. According to a report by the Orthopedic Device Regulatory Authority, nearly 30% of submissions fail due to inadequate quality management practices. This indicates a significant area for improvement within the industry.
Furthermore, ongoing monitoring post-market is crucial. Studies reveal that up to 20% of orthopedic implants may face complications over time. Proper quality management can minimize these risks. Continuous audits and feedback systems are necessary. However, some companies still struggle to implement effective practices. They may overlook the importance of user feedback. This gap can lead to failures in product performance and patient satisfaction.
| Quality Management Aspect | Standard / Regulation | Impact on Implants | Measurement Method |
|---|---|---|---|
| Material Quality | ISO 13485 | Ensures biocompatibility and mechanical strength | Material specifications and tests |
| Process Validation | FDA 21 CFR Part 820 | Reduces variability and defects | Statistical process control |
| Risk Management | ISO 14971 | Identifies and mitigates risks throughout lifecycle | Risk analysis and evaluation reports |
| Device Testing | ASTM Standards | Validates performance and safety of implants | Mechanical testing and fatigue testing |
| Supplier Management | ISO 9001 | Ensures consistency in component quality | Supplier audits and evaluations |
Quality management plays a crucial role in the success of orthopedic implants. Effective quality management practices can significantly reduce implant failure rates. When manufacturers implement stringent testing and quality checks, the risk of defects diminishes. This leads to improved durability and performance of orthopedic devices. Clinicians and patients often experience favorable outcomes when quality management is prioritized.
Patient outcomes are directly influenced by the reliability of the implants they receive. High-quality implants often result in shorter recovery times and fewer complications. However, not all manufacturers adhere to the same standards. Some may overlook essential quality checks, leading to potential risks. This inconsistency can create challenges for healthcare providers when selecting implants. The emphasis on quality management must remain a focal point for the industry.
Enhancing quality management involves continuous improvement and feedback. Manufacturers need to recognize that not all products will perform perfectly, and learning from failures is key. Understanding the root causes of implant failures can lead to better designs. Collaboration among stakeholders, including surgeons, patients, and manufacturers, is essential. Each party has insights that can help refine quality processes.
Quality management in orthopedic device manufacturing significantly influences financial outcomes. A recent report by the Orthopedic Industry Association highlights that implementing robust quality management systems can reduce production costs by up to 15%. This reduction stems from decreased waste, fewer product recalls, and lower risk of litigation. Improved quality control reduces the likelihood of defective products reaching the market.
Furthermore, studies show that organizations prioritizing quality management can enhance their market share. According to the International Standards Organization, companies with certified quality systems report a 20% increase in customer satisfaction. Satisfied customers tend to make repeat purchases, enhancing revenue streams. However, not all organizations realize these benefits. Some struggle with the cultural shift needed for effective quality management. Resistance to change can hamper progress.
Quality management is not just about compliance. It requires ongoing commitment and adaptation. Manufacturers must invest in employee training and incorporate feedback loops for continuous improvement. While the return on investment can be substantial, initial costs and possible disruptions must be considered. Balancing immediate expenses against long-term benefits becomes essential in decision-making. Quality management remains a vital consideration amid the complexities of orthopedic implant production.
This chart illustrates the cost-benefit analysis of implementing quality management practices in orthopedic device manufacturing. It compares the reduction in costs and improvement in patient outcomes as a result of enhanced quality management over a period of five years.
Quality management plays a crucial role in the development of orthopedic implants. One notable case study highlighted by the American Society for Quality (ASQ) showed that implementing Six Sigma methodologies reduced defect rates in implant production by 30%. This improvement was attributed to rigorous testing and quality assurance protocols. Surgeons reported better outcomes due to increased implant reliability.
Another example comes from a recent analysis by the Food and Drug Administration (FDA). It revealed that companies with strong quality management systems had 40% fewer recalls. These firms adhered to strict compliance guidelines and emphasized continuous training for their teams. However, not all companies are achieving such results. There are still significant gaps in documentation and processes that leave room for improvement.
While many organizations strive for excellence, some fail to meet industry benchmarks. The Orthopedic Research Society noted that only about 25% of new devices meet post-market surveillance criteria successfully. This highlights the need for a more robust quality management approach. It is essential to recognize both successes and setbacks. Through reflection, the industry can continue to advance the effectiveness of orthopedic implants.
: Quality management reduces implant failure rates by ensuring thorough testing and quality checks.
High-quality implants lead to shorter recovery times and fewer complications for patients.
No, some manufacturers overlook essential quality checks, creating potential risks in implant reliability.
Continuous improvement and feedback from stakeholders can enhance quality management practices.
Case studies showcase successful quality management practices and highlight areas needing improvement.
Companies with strong quality systems experienced 40% fewer recalls and better compliance.
No, only about 25% of new devices meet post-market surveillance criteria adequately.
Learning from failures can inform better designs and enhance overall implant quality.
Collaboration provides diverse insights to refine quality processes for more reliable implants.
The industry must recognize both successes and setbacks to advance orthopedic implant effectiveness.
Quality management plays a crucial role in the production of orthopedic implants, ensuring that they meet stringent standards and regulations. It involves the implementation of systematic processes that monitor and enhance quality throughout the manufacturing cycle. By adhering to established guidelines, manufacturers can significantly reduce the risk of implant failures and improve patient outcomes, leading to safer surgical experiences.
Furthermore, a cost-benefit analysis reveals that investment in quality management not only minimizes potential liabilities associated with faulty implants but also enhances overall operational efficiency. Case studies have demonstrated successful applications of quality management practices, showcasing improved performance and higher patient satisfaction. Therefore, understanding "what is the role of quality management in orthopedic implants" is essential for manufacturers aiming to deliver reliable and effective medical devices.

 XOVEC Orthopedic Implants
XOVEC Orthopedic Implants